A rare case of bacterial infective endocarditis caused by Streptococcus alactolyticus
CASE REPORT
A rare case of bacterial infective endocarditis caused by Streptococcus alactolyticus
Article Summary
- DOI: 10.24969/hvt.2019.133
- Page(s): 109-113
- CARDIOVASCULAR DISEASES
- Published: 07/06/2019
- Received: 30/05/2019
- Accepted: 07/06/2019
- Views: 14457
- Downloads: 8577
-
Citations
- Keywords: Streptococcus alactolyticus, endocarditis
PDF PRINT VERSION CommentsAddress for CorrespondenceAddress for Correspondence: Oben Baysan, Guven Hospital, Simsek Street No:29 Kavaklidere, Ankara, Turkey Mobile: +905322718225, Email: obbaysan@gmail.com
Nedim Cekmen, *Oben Baysan, Emine Disbudak, Ceren Gunt
Intensive Care Unit, *Cardiology Department, Guven Hospital, Ankara, TurkeyAbstract
Background: Streptococcus alactolyticus is a rarely isolated bacterium, which classified under DNA cluster IV of the S. Bovis/S. equinus complex. Infections, especially infective endocarditis, caused by Strep. alactolyticus are very rare in humans.
Case Report: We describe a case of Strepotoccocus alactolyticus bacteriemia complicated by infective endocarditis. A 64-year-old male with a previous history of coronary artery bypass grafting applied to our cardiology outpatient clinic with complaints of dyspnea, fever, confusion and an apical holosystolic murmur. He was admitted to the intensive care unit. Transthoracic and transesophageal echocardiography showed the presence vegetation on the aortic valve. S. alactolyticus was detected on serial blood cultures. The patient was first treated with intensive antimicrobial therapy, and then underwent mitral and aortic valve replacements with uneventful follow-up.
Conclusion: Streptococcus alactolyticus infective endocarditis has only been reported previously in one patient. More information is certainly needed for diagnosis and treatment of patients infected with Streptococcus alactolyticus.
Key words: Streptococcus alactolyticus, endocarditis
IntroductionStreptococcus alactolyticus is a particular subspecies grouped under streptococcal bovis/Streptococcal equinus complex (SBSEC) (1, 2). This complex contains non-beta hemolytic streptococcus and Lancefield group D streptococci. They are commensal colonizers of human and animal gastrointestinal tract and act as opportunistic pathogens (3).
S. alactolyticus has been isolated from intestinal flora of pigs, chicken, cows, pigeons and dogs (4, 5). Human infections caused by S. alactolyticus are extremely rare. Herein, we describe a rare case of bacterial infective endocarditis (IE) caused by S. alactolyticus.
Case report
A 64-year-old male was transferred to our emergency service by his family with difficulty of breathing, tachycardia, fever and confusion. He had previous history of coronary artery bypass graft surgery one month before. On admission, his vital signs were as follows high respiratory rate (32 breaths/min), temperature 38.6 C°, blood pressure 150/90 mmHg, heart rate 132 beats/min, and SpO2 67 %. He was urgently transferred to the intensive care unit (ICU). He had diminished respiratory sounds with crepitating rales bilaterally. We detected a grade 3/6 an apical holosystolic murmur on his left precordium. Glasgow coma score was 9, Acute Physiology and Chronic Health Evaluation score (APACHE II) was 18 and his Multiple Organ Dysfunction Score (MODS) was 6. Arterial blood gas analysis showed metabolic acidosis and hypoxemia: (bicarbonate 17.2 mmol/L, pH 7.14, PaCO2 35 mm Hg, and PaO2 47 mm Hg while on nasal oxygen at 6 L/min).
Blood cultures were taken. He was intubated and mechanically ventilated. Laboratory analyses showed high leukocyte count (23.400/L) with neutrophil dominance (83%), erythrocyte sedimentation rate 62 mm/h, C-reactive protein 135 mg/L (reference range: 0-5 mg/L) , procalcitonin 2.4 ng/L, and creatinine 1.4 mg/dL Immediate bedside transthoracic echocardiography revealed a suspicious mass on the aortic valve with severe aortic regurgitation. There was also a moderate degree mitral regurgitation. Same day urgent transesophageal echocardiogram (TEE) confirmed presence of a large mobile vegetation (23x15 mm) on aortic valve associated with severe aortic regurgitation (Fig. 1 and 2) and moderate degree mitral regurgitation (Fig. 3). Empiric antimicrobial therapy with vancomycin, gentamicin, and penicillin G was initiated. His general condition showed a noticeable improvement within 48 hours. The group D streptococci were identified from blood cultures as S. alactolyticus by Vitek 2 (bioMerieux Vitek, Inc, Hazelwood, St. Louis, MO, USA). It was susceptible to penicillin. The second set of blood cultures were also positive for S. alactolyticus. Vancomycin was discontinued and intravenous penicillin G (3. 000.000 U every 4 hr) and gentamicin (63 mg every 8 hr) were continued. With that therapy, he showed persistent clinical improvement. His subsequent blood cultures were negative. The patient was extubated on the 10th day of his intubation. The neurologist’s consultation including brain magnetic resonance imaging (MRI) was unremarkable for the presence of cerebral embolism. We recommended to the patient early surgical intervention. He agreed with the proposal and underwent valvular heart surgery with bioprosthetic aortic and mitral valve replacements. Based on the S. alactolyticus sensitivities, antimicrobial therapy was deescalated to ceftriaxone, which was planned to continue for another six-weeks. The patient recovered completely and was discharged on the 44th day of hospitalization. He was well on follow-up 8 weeks after discharge. He was also informed about an outpatient gastroenterology consultation for possible colonoscopy procedure.
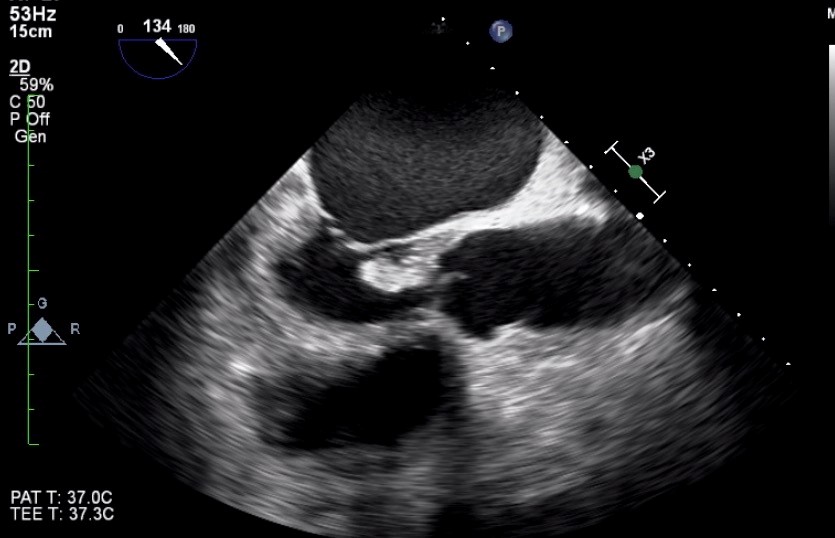
Figure 1. A large aortic valve vegetation on TEE image (Mid esophageal 134 degree)
TEE- transesophageal echocardiography
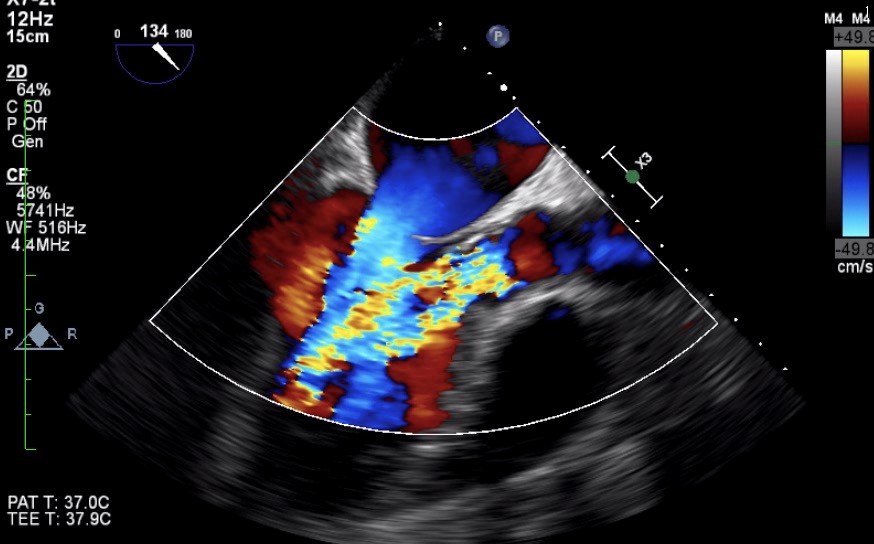
Figure 2. Severe aortic regurgitation caused by infective endocarditis.
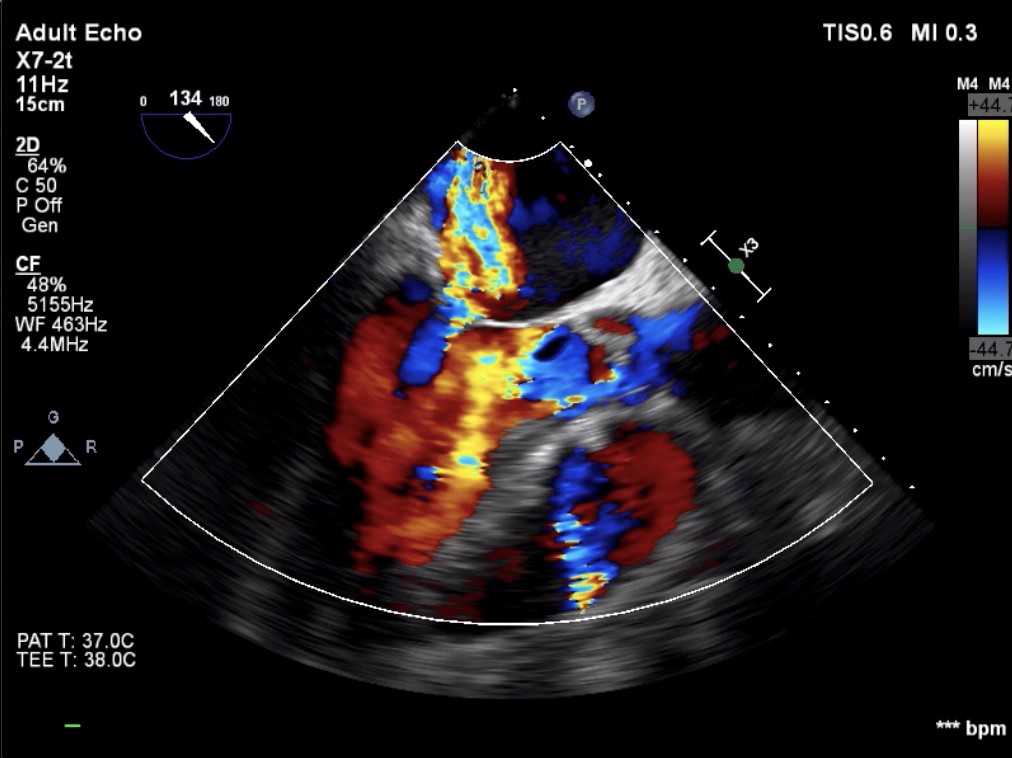
Figure 3. Moderate mitral regurgitation
Discussion
S. Bovis/S. equinus complex bacteria contains both good strains such as S. gallolyticus subsp. macedonicus and S. lutetiensis ingested in daily food products and pathogenic strains (6). They inhabit gastrointestinal tract of animals and humans (7). Colon and ileum are major colonization sites for S. Bovis/S. equinus complex and they have a well-known association with colorectal cancer (8, 9). Bacteremia and infective endocarditis are frequently encountered diseases for human SBSEC infection.
Geographical location and development status of a country determine distribution of main pathogens causing IE. Indeed, Staphylococcus aureus is still the most frequently detected bacteria for all IE cases in developed countries (31%) followed by viridans group streptococci (17%), coagulase negative staphylococci (11%), enterococci (10%) and finally S. Bovis/S. equinus complex (SBSEC) (6%) (10). In contrast, Streptococcus still predominates in low to middle income countries (11, 12). Interestingly, the percentage of SBSEC-associated IE within all cases of streptococcal IE increased from 10.9% to 23.3% from 1995 to 2005 (6). Within SBSEC bacteria S. gallolyticus subsp. gallolyticus is a most prominent agent in IE cases across Europe. Another bacteria in this complex, S. gallolyticus subsp. Pasteurianus, is also the leading agent in Asia (13).
Human reports of S. alactolyticus infection are exceedingly rare. S. alactolyticus was reported as a causative agent in a patient with IE complicated by septic emboli (14). Another fatal case of fulminant neonatal sepsis caused by this pathogen was also reported (15).
Our patient presented with S. alactolyticus bacteriemia and aortic valve endocarditis. Aortic valve is the least frequently involved heart valve in SBSEC IE (16). In contrast, there is a tendency for SBSEC bacteria to affect multiple heart valves including mitral and prosthetic valves (16). The reported frequency of embolic events in SBSEC IE ranged from 9% to 55% (17, 18). Fortunately, the patient in our case had no embolic event confirmed by a neurologic exam and brain MRI. That could be related to relatively early diagnosis and prompt starting of antimicrobial therapy.
Conclusion
There is unmet need for better clarification of infective endocarditis caused by SBSEC bacteria. An umbrella term SBSEC contains many subspecies. The new SBSEC taxonomical grouping (1) brings new challenges and opportunities to clinicians. More reliable estimates of SBSEC related IE frequency and incidence could be obtained via the correct identification of SBSEC bacteria in each IE case, which provides better clarification of SBSEC IE epidemiology.
Peer-review: internal and external
Conflict of interest: None to declare
Authorship: N.C., O.B., E.D., C.G. equally contribute to case management and preparation of case report
Acknowledgement and funding: None to declare
References
1. Schlegel L. Reappraisal of the taxonomy of the Streptococcus bovis/Streptococcus equinus complex and related species: description of Streptococcus gallolyticus subsp. gallolyticus subsp. nov., S. gallolyticus subsp. macedonicus subsp. nov. and S. gallolyticus subsp. pasteurianus subsp. nov. Int J System Evolut Microbiol 2003; 53 :631. https://doi.org/10.1099/ijs.0.02361-0 PMid:12807180 2. Facklam R. What happened to the streptococci: overview of taxonomic and nomenclature changes. Clin Microbiol Rev 2002;15: 613. https://doi.org/10.1128/CMR.15.4.613-630.2002 PMid:12364372 PMCid:PMC126867 3. Dekker JP, Lau AF, Kraft CS. An update on the Streptococcus bovis group: classification, ıdentification, and disease associations. J Clin Microbiol 2016; 54: 1694. https://doi.org/10.1128/JCM.02977-15 PMid:26912760 PMCid:PMC4922088 4. Farrow JAE, Kruze J, Phillips BA, Bramley A, Collins MD. Taxonomic studies on Streptococcus bovis and Streptococcus equinus: Description of Streptococcus alactolyticus sp. nov. and Streptococcus saccharolyticus sp. nov. System Appl Microbiol 1984;5:467-82. https://doi.org/10.1016/S0723-2020(84)80004-1 5. Rinkinen ML, Koort JMK, Ouwehand AC, Westermarck E, Björkroth KJ. Streptococcus alactolyticus is the dominating culturable lactic acid bacterium species in canine jejunum and feces of four fistulated dogs. FEMS Microbiol Letters 2004; 230: 35-9. https://doi.org/10.1016/S0378-1097(03)00851-6 6. Jans C, Meile L, Lacroix C, Stevens MJA. Genomics, evolution, and molecular epidemiology of the Streptococcus bovis/Streptococcus equinus complex (SBSEC). Infection, genetics and evolution. J Mol Epidemiol Evolut Genet Infect Dis 2015; 33: 419-36. https://doi.org/10.1016/j.meegid.2014.09.017 PMid:25233845 7. Jans C, Boleij A. The road to infection: host-microbe interactions defining the pathogenicity of Streptococcus bovis/Streptococcus equinus complex members. Front Microbiol 2018; 9: 603.https://doi.org/10.3389/fmicb.2018.00603 PMid:29692760 PMCid:PMC5902542 8. Dahmus JD, Kotler DL, Kastenberg DM, Kistler CA. The Gut microbiome and colorectal cancer: a review of bacterial pathogenesis. J Gastrointest Oncol 2018; 9: 769-77. https://doi.org/10.21037/jgo.2018.04.07 PMid:30151274 PMCid:PMC6087872 9. Boleij A, Roelofs R, Schaeps RMJ, Schülin T, Glaser P, Swinkels DW, et al. Increased exposure to bacterial antigen RpL7/L12 in early stage colorectal cancer patients. Cancer 2010; 116: 4014-22.https://doi.org/10.1002/cncr.25212 PMid:20564125 PMCid:PMC2930125 10. Murdoch DR, Corey GR, Hoen B, Miró JM, Fowler VG, Bayer AS, et al. Clinical presentation, etiology, and outcome of infective endocarditis in the 21st century: the International Collaboration on Endocarditis-Prospective Cohort Study. Arch Intern Med 2009; 169: 463-73. https://doi.org/10.1001/archinternmed.2008.603 PMid:19273776 PMCid:PMC3625651 11. Mirabel M, Rattanavong S, Frichitthavong K, Chu V, Kesone P, Thongsith P, et al. Infective endocarditis in the Lao PDR: clinical characteristics and outcomes in a developing country. Int J Cardiol 2015;180:270‐3.https://doi.org/10.1016/j.ijcard.2014.11.184 PMid:25482077 PMCid:PMC4323144 12. Math RS, Sharma G, Kothari SS, Kalaivani M, Saxena A, Kumar AS,et al. Prospective study of infective endocarditis from a developing country. Am Heart J 2011; 162: 633‐8. https://doi.org/10.1016/j.ahj.2011.07.014 PMid:21982654 13. Lee RA, Woo PCY, To APC, Lau SKP, Wong SSY. Geographical difference of disease association in Streptococcus bovis bacteraemia. J Med Microbiol 2003; 52: 903-8. https://doi.org/10.1099/jmm.0.05199-0 PMid:12972586 14. Almeida P, Railsback J, Gleason JB. A rare case of Streptococcus alactolyticus ınfective endocarditis complicated by septic emboli and mycotic left middle cerebral artery aneurysm. Case Rep Infect Dis 2016; 2016: 9081352. https://doi.org/10.1155/2016/9081352 PMid:27525136 PMCid:PMC4972922 15. Toepfner N, Shetty S, Kunze M, Orlowska-Volk M, Krüger M, Berner M, et al. Fulminant neonatal sepsis due to Streptococcus alactolyticus - A case report and review. APMIS 2014; 122: 654-6.https://doi.org/10.1111/apm.12219 PMid:24475920 16. Hoen B, Chirouze C, Cabell CH, Selton-Suty C, Duchêne F, Olaison L, et al. Emergence of endocarditis due to group D streptococci: findings derived from the merged database of the International Collaboration on Endocarditis. Eur J Clin Microbiol Intect Dis 2005; 24: 12-6. https://doi.org/10.1007/s10096-004-1266-6 PMid:15660254 17. Kupferwasser I, Darius H, Müller AM, Mohr-Kahaly S, Westermeier T, Oelert H, et al. Clinical and morphological characteristics in Streptococcus bovis endocarditis: a comparison with other causative microorganisms in 177 cases. Heart 1998; 80: 276-80. https://doi.org/10.1136/hrt.80.3.276 PMid:9875088 PMCid:PMC1761108 18. Pergola V, Di Salvo G, Habib G, Avierinos JF, Philip E, Vailloud JM, et al. Comparison of clinical and echocardiographic characteristics of Streptococcus bovis endocarditis with that caused by other pathogens. Am J Cardiol 2001; 88: 871-5. https://doi.org/10.1016/S0002-9149(01)01914-2 Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.Archive of Issues
AUTHOR'S CORNER
 Authors having problems with submissions please notify editor: editor@hvt-journal.com
Authors having problems with submissions please notify editor: editor@hvt-journal.com
