The effect of progesterone on cardiomyocytes in traumatic brain injury: A randomized study
ORIGINAL RESEARCH ARTICLE
The effect of progesterone on cardiomyocytes in traumatic brain injury: A randomized study
Article Summary
- DOI: 10.24969/hvt.2025.610
- CARDIOVASCULAR DISEASES
- Published: 30/11/2025
- Received: 26/09/2025
- Revised: 11/10/2025
- Accepted: 11/10/2025
- Views: 1397
- Downloads: 1042
- Keywords: Traumatic brain injury, cardiomyocytes, correction, progesterone, rats, randomized study
Address for Correspondence: Mariia S. Shuvalova, Saint Petersburg State Pediatric Medical University, Saint Petersburg, Russia
Email: shuvalova.88@internet.ru
ORCID: Mariia S. Shuvalova - 0000-0002-2295-090X; Sergey A. Lytaev - 0000-0002-1291-0381
Mariia S. Shuvalova, Sergey A. Lytaev
Saint Petersburg State Pediatric Medical University, Saint Petersburg, Russia
Abstract
Objective: Traumatic brain injury (TBI) is one of the leading causes of death and disability in the world, especially among young people of working age. In addition to direct brain damage, TBI causes repercussive organ changes, including dysfunction of the cardiovascular system, which worsens the prognosis of patients. Currently, there are no effective medicines for the treatment of TBI, and the search for new neuroprotective and cardioprotective drugs remains an urgent task. Progesterone, widely used in gynecology, has demonstrated neuroprotective properties in experimental models of TBI, but its effect on the heart during trauma has not been sufficiently studied.
The aim of the study was to study the effect of progesterone on the morphological state of cardiomyocytes in acute TBI.
Methods: The experiment was conducted on 15 sexually mature outbred male rats (200-250 g), divided into three groups of 5 individuals: intact (control), with simulated acute TBI without treatment, and with TBI treated with progesterone (10 mg/kg 1 and 24 hours after injury). The closed TBI model was reproduced by the fall of a metal weight (68 g) from a height of 1 m, creating an impact force of 0.6 J. On the 3rd day after the injury, the animals were removed from the experiment and a morphological analysis of the heart was performed. Myocardial samples were fixed, stained, and studied under a light microscope with cardiomyocyte morphometry (thickness, area, nuclear-cytoplasmic ratio, pycnotic index). Statistical data processing was performed using SPSS 16.0, the Student's t-test was applied at p < 0.05.
Results: On the 3rd day after the induction of moderate TBI cardiomyocytes exhibited changes including loss of their cylindrical shape, disruption of transverse striation, cytoplasmic vacuolization, as well as signs of apoptosis and necrosis. Cell nuclei were deformed, hyperchromatic, and showed karyorrhexis, accompanied by a significant reduction in morphometric parameters of cardiomyocytes: cell length decreased from 95.2 (4.8) to 73.6 (3.9) μm, cross-sectional diameter decreased by 23%, and cross-sectional area decreased by 28% compared to the control group (p<0.05). Nuclear diameter decreased from 7.2 (0.5) to 5.3 (0.4) μm, and the nuclear-cytoplasmic ratio fell by 27%.
Progesterone administration contributed to partial restoration of morphometric parameters: cardiomyocyte length increased to 85.7 (4.1) μm, and cross-sectional diameter to 12.6 (1.0) μm. The frequency of pyknotic nuclei decreased, and the number of necrotic cells was reduced. Morphologically, preservation of transverse striation, orderly arrangement of cardiomyocytes, and diminished signs of inflammation were observed.
Conclusion: The data obtained indicate the ability of progesterone to partially restore the structure of the heart muscle and reduce degenerative processes, which opens up prospects for complex therapy of patients with TBI.
Key words: Traumatic brain injury, cardiomyocytes, correction, progesterone, rats, randomized study
Introduction
Traumatic brain injury (TBI) remains a serious medical and social problem. Today, traumatic injury occupies the third place in the structure of mortality of the world's population, second only to cardiovascular and oncological diseases (1), while being the leading cause of disability and mortality among young people in developed countries (2). Thus, in 2019, 27.16 million new cases of TBI were registered worldwide, of which falls were the main cause in 74% of cases, pedestrian traffic injuries accounted for 14%, motor vehicle traffic injuries 5%, and conflicts and terrorism accounted for 3% of the total causes of TBI (3).
In Russia, about 600 thousand people per year receive TBI, of which about 50 thousand are fatal or acquire permanent disability (4).

It should be noted that the most numerous group of TBI remains young men of working age, but the prognosis for older people over 60 years is worse and is characterized by the highest mortality rate (5). The high prevalence of the problem, the high cost of treatment, and the large number of cases that lead to disability of the able-bodied population make the problem of TBI a heavy burden not only for an individual or a family, but also an important economic problem for the state. Hence, the search for modern treatment methods that will improve the outcome of TBI is a priority in modern medicine.
The mechanism of TBI development is complex and all the long-term consequences of this condition are not fully understood. In the light of modern concepts, TBI is considered as one of the triggers for the development of diseases of the cardiovascular system (6). Thus, in retrospective studies conducted 6 months after TBI, it was proved that TBI was an independent risk factor for the development of myocardial dysfunction characterized by cardiac arrhythmia, coronary heart disease and heart failure (7). The described phenomenon may be associated with nonspecific changes in the endothelium in critical conditions, which can form in the microcirculatory bed of the heart, causing organ dysfunction (8). Circulatory insufficiency in patients with concomitant TBI develops already on the first day of the post-traumatic period due to a decrease in the contractile function of the heart and is an important extracranial factor of secondary brain damage (9). Therefore, the urgent task of treating a traumatic illness is the timely correction of microcirculation disorders not only in the brain, but also in the heart muscle.
To date, there is no effective drug treatment for TBI. The main approach is supportive: intracranial pressure monitoring, ventilation, and neurorehabilitation. Therefore, the search and development of neuroprotective drugs capable of affecting individual links of pathogenesis and reducing brain damage after injury remains the main priority of medicine.
Despite the fact that progesterone is one of the most popular endogenous steroids and progestogenic sex hormones in gynecological practice with many years of experience, in the 1990s, Stein et al. discovered its neuroprotective properties in experimental models of brain injury (10). A group of scientists conducted studies that showed that the introduction of progesterone reduces brain edema, protects neurons, and improves functional outcome in the experiment (10). However, to date, the issue of the extracerebral effects of progesterone in TBI has not been fully studied.
Therefore, the purpose of this study was to determine the effect of progesterone on the state of cardiac cardiomyocytes in acute traumatic brain injury.
Methods
Study design and animals
An experimental randomized study was conducted on 15 sexually mature outbred male rats weighing 200-250 g, obtained from the vivarium of the Medical Faculty of the Kyrgyz-Russian Slavic University. The animals were kept in standard conditions: at a temperature of 21-23 °C, 12-hour light mode, with free access to water and food. The study was conducted in accordance with the laboratory standards approved by the Order of the Ministry of Health and Social Development of the Russian Federation dated August 23, 2010 № 708 N "On approval of the rules of laboratory practice." and international standards for animal studies. The research protocol was approved by the local ethics committee at the NGO "Preventive Medicine" of the Ministry of Health of the Kyrgyz Republic (Protocol № 1 dated 5.01.2020).
Graphical abstract
The animals were randomly divided into3 groups of 5 individuals each: group 1 (control) (n=5) consisted of intact rats without TBI; group 2 (n=5) included rats with simulated acute TBI without subsequent treatment; group 3-the 1st group (n=5) consisted of rats with simulated TBI treated with progesterone.
Experiment
The model of closed craniocerebral injury was reproduced according to the method described by Tang et al. (1997), with adaptations to the laboratory base used and the weight category of animals.
A metal weight (68g) enclosed in a tube with a diameter of 2 cm by free fall from a height of 1 m struck a rod through which energy was transferred to the animal's head. The impact force of 0.6 joules was transmitted to the skull bones and underlying tissues of the fixed animal, simulating a closed injury of moderate severity. The assessment of the degree of TBI was confirmed by neurological tests: the open field method, the rake test and the somatosensory test.
After injury, the animals were transferred to individual cages until independent breathing and motor activity were restored, general activity, seizures, hemorrhages, etc. were recorded.
Animals of the 3rd group (n=5) were injected with progesterone at a dose of 10 mg / kg body weight 1 hour and 24 hours after the application of TBI.
On the 3rd day after the injury, the animals of all three groups were removed from the experiment using an overdose of chloroform. The heart was taken and fixed for morphological analysis. Myocardial samples were fixed in 10% neutral formalin, embedded in paraffin, thin sections were made, stained with hematoxylin and eosin, as well as Van Gieson for connective tissue assessment. The material for the study was the myocardium of the heart. The thickness of cardiomyocytes, the density of the vascular network, the degree of stasis and edema were evaluated.
The finished histological preparations were studied using an Olympus BX40 light microscope (Japan) at magnifications of ×100 and ×400. At the same time, photographic documentation of the studied areas was carried out using a Levenhuk C130 NG digital camera (PRC) connected to a microscope, followed by image transfer to a personal computer. All observations and morphological changes were recorded during the study.
Morphometric analysis was performed using standard software integrated into the Olympus СellSens microscopic system. The length (CL), diameter (DC), and cross-sectional area of cardiomyocytes (CCS), the diameter of the nucleus (DN), the nuclear-cytoplasmic ratio (NCR), and the pycnotic nucleus index (PN) were determined.
The cross-sectional area of cardiomyocytes was calculated using the formula
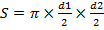 ,
,
where
S – is the cross–sectional area of the cardiomyocyte,
d1 – is the width of the cardiomyocyte,
d2 – is the thickness of the cell.
The nuclear-cytoplasmic ratio (NCR) of the cell was estimated by the formula:
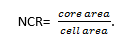
The pycnotic nuclei were counted using the visual method in 10 random fields of view of a microscope, counting the total number of nuclei, as well as the number of pycnotic nuclei, followed by determining the percentage of the latter.
Statistical analysis
Quantitative results of the morphometric analysis were processed using the SPSS 16.0 statistical software package. A non-parametric analysis method was chosen due to the small sample sizes. The Mann–Whitney U test was used for comparisons between two groups. The Kruskal–Wallis test was applied to compare three groups, and for post-hoc pairwise comparisons between groups, the Mann–Whitney U test was again used. Additionally, the parametric Student's t-test was employed to evaluate the statistical significance of differences between groups. Differences were considered statistically significant at a p-value of less than 0.05 (p < 0.05).
Results
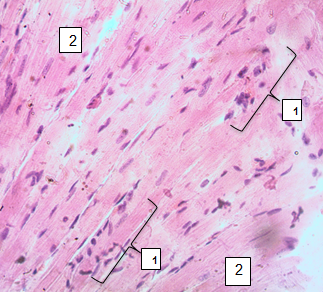
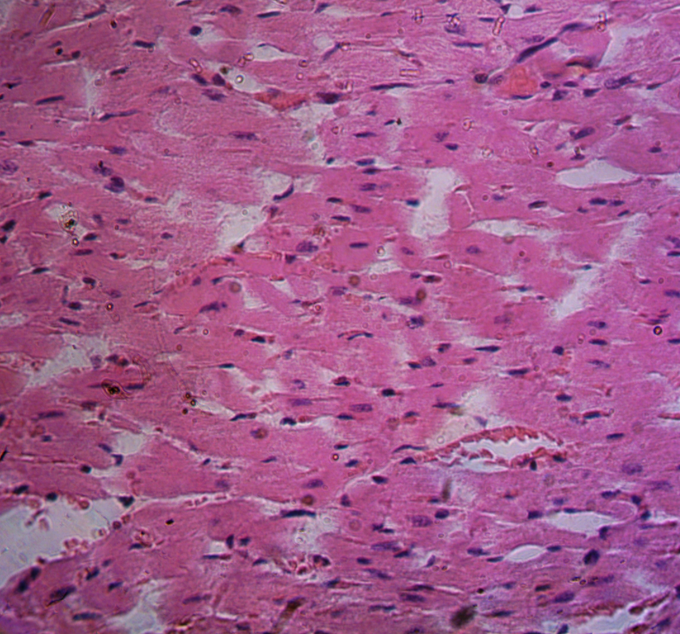
On the 3rd day after a moderate TBI, cardiomyocytes undergo a number of destructive and degenerative changes (Fig.1) compared to the control group (Fig. 2). The cells lose their usual cylindrical shape, become unevenly thickened, with areas of cytoplasmic vacuolization. The transverse striation is partially or completely absent, especially in the peripheral areas.
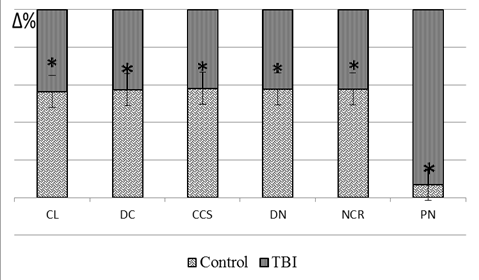
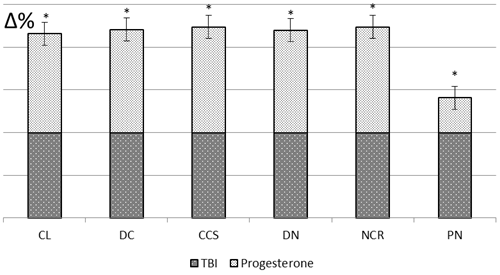
The light microscopy data is consistent with the results of the morphometric study (Table 1, Fig. 3). Modulations in the structural organization of cardiomyocytes are observed in animals with TBI on the 3rd day of the experiment. Thus, the length of cardiomyocytes, normally 95.2 (4.8) microns, decreases to 73.6 (3.9) microns (p<0.05), while the transverse diameter decreases by 23% in the second group (TBI) compared with the control group (p < 0.05). The cross-sectional area of the cells decreases by 28% (p<0.05) of the initial size (p<0.05).
A decrease in the length and transverse diameter of cardiomyocytes, in our opinion, may indicate the initial stages of the development of atrophic changes and a decrease in the functional activity of the heart muscle due to hypoxia, metabolic disorders and neurovegetative shifts that occur during TBI.
|
|
|
|
Figure 1. Rat myocardium on the 3rd day after traumatic brain injury, stained with hemotoxylin-eosin, ×400. 1 – compressed, hyperchromic nucleus (pyknosis) with elements of fragmentation (karyorexis) 2 – areas of necrotic cells |
Figure 2. Cardiomyocytes of an intact rat, stained with hemotoxylin-eosin, ×400.
|
|
Table 1. Comparative morphometric parameters of cardiomyocytes |
|||
|
Variables |
Group1 (control) (n=5) |
Group 2 (TBI) (n=5) |
Group 3 (TBI + progesterone) (n=5) |
|
Cardiomyocyte length, microns |
95.2 (4.8) (85.6 – 104.8) |
73.6 (3.9)* (65.8 – 81.4) p < 0.05 |
85.7 (4.1)# (77.5 – 93.9) p < 0.05 |
|
Diameter of a cardiomyocyte, microns |
14.1 (1.2) (11.7 – 16.5) |
10.4 (1.1)* (8.2 – 12.6) p < 0.05 |
12.6 (1.0)# (10.6 – 14.6) p < 0.05 |
|
Cross-sectional area, microns² |
170.3 (9.5) (151.3 – 189.3) |
122.7 (11.2)* (100.3 – 145.1) p < 0.001 |
152.4 (8.7)# (135.0 – 169.8) p < 0.001 |
|
Diameter of the cardiomyocyte nucleus, µm |
7.2 (0.5) (6.2 – 8.2) |
5.3 (0.4)* (4.5 – 6.1) p < 0.05 |
6.4 (0.3)# (5.8 – 7.0) p < 0.05 |
|
Nuclear-cytoplasmic relationship |
0.52 (0.03) (0.46 – 0.58) |
0.38 (0.04)* (0.30 – 0.46) p < 0.05 |
0.47 (0.03)# (0.41 – 0.53) p < 0.05 |
|
Pycnotic nucleus (per 100 cells),% |
3.1 (0.8) (1.5 – 4.7)
|
42.5 (4.3)* (33.9 – 51.1) p < 0.05 |
17.3 (2.5)# (12.3 – 22.3) p < 0.05 |
|
Data are presented as Mean (SD) and range? Kruskal - Wallis test, post test Mann Whitney U test *Significant differences were observed in the group with TBI compared to the control group (p < 0.05) # Significant differences in the outcomes were found between the group with correction and the group without correction using the drug (p < 0.05) TBI – traumatic brain injury |
|||
|
|
|
Figure 3. Morphometric analysis of the main characteristics of cardiomyocytes in animals that have suffered traumatic brain injury (%) CL - cardiomyocyte length, DC - diameter of a cardiomyocyte, CCS - cardiomyocyte cross-sectional area, DN - diameter of the cardiomyocyte nucleus, NCR - nuclear-cytoplasmic relationship, PN - pycnotic nucleus, TBI – traumatic brain injury * — significant differences from the control group (p < 0.05) |
The nuclear apparatus of the cardiomyocyte of the heart of an intact animal is represented by one or two oval, light nuclei located in the center, equipped with clearly distinguishable nucleoli, which corresponds to a high level of metabolic activity. In TBI, karyopycnosis, karyorexis, and markers of apoptosis are observed in a number of cells, indicating activation of cell death. The nuclei are deformed, displaced, and often hyperchromic or completely lysed (Fig. 1). Morphometry determines a decrease in the diameter of cardiomyocyte nuclei from 7.2 (0.5) microns in the control group to 5.3 (0.4) microns (p<0.05) in the group of animals with TBI. The cytoplasm acquires a granular and uneven oxyphilic character, which indicates a violation of organellar integrity (Fig. 1).
The nuclear-cytoplasmic ratio in cardiomyocytes during TBI decreases by 27% (p<0.05) compared with the control group (Fig. 3). Clusters of fragmented and necrotized cardiomyocytes are recorded in separate fields of vision, mainly in the subendocardial and subepicardial sections of the myocardium (Fig. 1). Visually, nuclear pyknosis is clearly visible in the field of view of the microscope. Pycnotic nuclei occur in 42.5 (4.3)% of the total number of nuclei in histological sections of the myocardium of rats with TBI, whereas in the control group this indicator is only 3.1 (0.8)% (p<0.05), which indicates a significant increase in the frequency of nuclear changes associated with apoptosis or necrosis.
Against the background of progesterone administration, there is a partial restoration of the structure of cardiomyocytes and a decrease in the severity of degenerative processes: cardiomyocytes of regular elongated shape predominate, while maintaining transverse striation. The cell nuclei are mostly centrally located, oval in shape, with light chromatin and preserved nucleoli. Signs of apoptosis (karyopycnosis, karyorexis) are much less common than in the TBI group without correction, and there are no rough areas of coagulation necrosis. Signs of moderate cytoplasmic vacuolation persist in some visual fields, which may reflect residual effects of cellular stress.
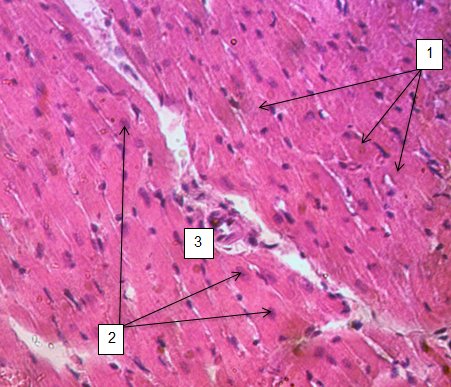
Morphoscopic examination of the myocardium of rats who had suffered moderate TBI and received progesterone as a correction for this condition revealed moderate changes (Fig. 4), differing in severity from the group without treatment. Thus, the structure of cardiomyocytes is preserved, the cells are arranged in an orderly manner, their contours are clear, and the transverse striation is predominantly preserved.
|
|
|
Figure 4. Rat myocardium on the 3rd day after TBI, receiving progesterone correction, hematoxylin-eosin staining, ×400 1 – cardiomyocytes with moderately oxyphilic cytoplasm, clear contours; 2 – nuclei of cardiomyocytes with clearly distinguishable nucleoli; 3 – the artery of the heart TBI – traumatic brain injury
|
Positive changes in morphometric parameters of cardiomyocytes were revealed during progesterone
correction (Fig. 5).
|
|
|
Figure 5. Morphometric analysis of the main characteristics of cardiomyocytes of animals with TBI on the background of progesterone correction (%) CL - cardiomyocyte length, DC - diameter of a cardiomyocyte, CCS - cardiomyocyte cross-sectional area, DN - diameter of the cardiomyocyte nucleus, NCR - nuclear-cytoplasmic relationship, PN - pycnotic nucleus, TBI – traumatic brain injury * — significant differences from the control group (p < 0.05) |
Thus, the length of cardiomyocytes in animals with TBI treated with the drug was 85.7 (4.1) microns, which significantly exceeds the corresponding indicator in the TBI group without treatment (73.6(3.9) microns, p < 0.05). There was also an increase in the transverse diameter of the cells, reaching 12.6 (1.0) microns in the experimental group versus 10.4 (1.1) microns in the control (p<0.05). Moreover, the cross-sectional area of cardiomyocytes in the progesterone group increased by 24% compared with the intact group (p<0.05), which may indicate a decrease in the severity of dystrophic and atrophic processes in the myocardium under the influence of progesterone TBI correction.
The nuclei of cardiomyocytes are predominantly oval in shape, moderately hyperchromic, and centrally localized (Fig. 4). Morphometric analysis showed that the diameter of the nuclei significantly increased in the progesterone-corrected group and amounted to 6.4 (0.3) microns compared with 5.3 (0.4) microns in the TBI group without correction (p < 0.05; Fig. 5). At the same time, the nuclear-cytoplasmic ratio increases by 23% compared to the untreated group, which may indicate activation of nuclear activity and restoration of the metabolic status of the cell.
Single pycnotic nuclei are noted in individual cardiomyocytes, however, their number is significantly lower compared to the group of animals without correction and amounts to 17.3 (2.5)% versus 42.5 (4.3)%, respectively (p<0.05). In addition, there is a significant decrease in the number of fragmented and necrotic cells, mainly in the subendocardial and subepicardial sections of the myocardium. Signs of leukocyte infiltration are weak or absent, which further confirms the anti-inflammatory and cytoprotective effect of progesterone.
Discussion
In our study, it was found that TBI entails, among other things, extracranial phenomena, in particular, remodeling of cardiac cardiomyocytes.
Thus, TBI causes a systemic inflammatory reaction and the release of catecholamines, which leads to the release of neurotransmitters and cytokines such as interleukin-1, interleukin-6, interleukin-10 and tumor necrosis factor-α. The catecholamine storm and cytokine release secondary to TBI cause nonspecific and disseminated activation of the alpha and beta catecholamine receptors, which ultimately leads to impaired cardiac function and impaired regulation of cardiac contraction (14-16).
In turn, steroid hormones (estrogens, androgens, progestins, glucocorticoids, mineralocorticoids, and vitamin D) as universal biological regulators, they participate in the control of almost all physiological functions of the body, in particular, progesterone is an important neurosteroid that reduces oxidative stress during TBI, exhibits anti-inflammatory activity, stabilizes the blood-brain barrier, suppresses apoptosis and stimulates remyelination (14). Given the presence of common pathophysiological cascades in the development of repercussive changes in the heart muscle during TBI, the protective effects of progesterone may also be relevant for cardiomyocytes.
According to Dai et al. (17) progesterone receptors are present in cardiomyocytes, as well as the mitochondrial PR‑M receptor, whose stimulation increases β‑oxidation and mitochondrial metabolism in heart cells (H9c2). The improvement of myocardial metabolism against the background of progesterone use in rats explains the positive dynamics on the part of cardiomyocytes, namely the incomplete restoration of the structure of cardiomyocytes and the weakening of degenerative changes.
The nuclear apparatus of cardiomyocytes has a central location, oval-shaped nuclei containing light chromatin and visible nucleoli. In other words, in the group of rats treated with progesterone TBI, the apoptotic activity of cardiomyocytes decreases, and there are no rough zones of coagulation necrosis. This effect can be interpreted by reducing the activation of caspase‑3 and the release of cytochrome C by stimulating the expression of antiapoptotic Bcl‑xL, glutathione peroxidase, and other antioxidant enzymes (18). In some areas, moderate vacuolization of the cytoplasm persists, which may indicate the residual effects of cellular stress. In a model of myocardial infarction in adult mice, daily injection of progesterone improved the ejection fraction, reduced fibrosis, and increased proliferation in the injury area (19), which in our work led to an increase in the survival rate of animals.
The restoration of the myocardial structure, expressed in the predominance of regular elongated cardiomyocytes with preserved transverse striation and orderly arrangement of cells, indicates a reduction in the damaging effect of injury on the heart muscle. The central location of the nuclei, light chromatin and preserved nucleoli indicate the preservation of cell viability and a decrease in cellular stress. However, in the experiment with the use of progesterone, the positive dynamics is partial.
A significant decrease in signs of apoptosis, such as karyopycnosis and karyorexis, as well as the absence of coagulation necrosis sites in the progesterone group confirm its cytoprotective properties.
These data are consistent with previously published studies in which progesterone showed anti-inflammatory and anti-apoptotic effects in various models of tissue damage, including neuronal and muscular structures (20).
Nevertheless, the persistence of moderate cytoplasmic vacuolation in certain areas of the myocardium indicates the presence of residual cellular stress, which may reflect incomplete recovery or the need for longer therapy. This highlights the importance of further research aimed at optimizing the dosage and duration of progesterone administration, as well as studying its mechanisms of action at the molecular level.
Study limitations
The limitation of the study is the small number of animals included in investigation.
Conclusions
1. Traumatic brain injury leads to significant changes in the myocardium of the heart, namely dystrophic and necrobiotic changes in cardiomyocytes, leading to tissue hypoxia and ischemia, activation of apoptosis and inflammatory response in the heart tissue.
2. Prescribing progesterone to animals with TBI has a pronounced cardioprotective effect, which is confirmed by the morphological picture of the myocardium, which differs significantly from the changes observed in TBI without treatment.
3. Progesterone helps preserve the structural integrity of cardiomyocytes, reduces the severity of dystrophic and apoptotic processes in the myocardium, and helps stabilize the morphofunctional state of the myocardium, which indicates its cardioprotective potential in systemic pathology caused by TBI.
Ethics: The study was conducted in accordance with the laboratory standards approved by the Order of the Ministry of Health and Social Development of the Russian Federation dated August 23, 2010 № 708 N "On approval of the rules of laboratory practice." and international standards on animal studies. The research protocol was approved by the local ethics committee at the NGO "Preventive Medicine" of the Ministry of Health of the Kyrgyz Republic (Protocol № 1 dated 5.01.2020).
Peer-review: External and internal
Conflict of interest: None to declare
Authorship: M.S.S. and S.A.L equally contributed to the study and manuscript preparation , approved the final manuscript and
fulfilled the authorship criteria
Acknowledgements: The authors of the article express their special gratitude to Yu.Ch-M. Shidakov, head of the Laboratory of Experimental Process Modeling at the Faculty of Medicine of the Kyrgyz-Russian Slavic university, for providing a scientific platform for experiments and assistance in conducting them
Funding: None to declare
Statement on A.I.-assisted technologies use: Authors did not use A.I. technology in preparation of manuscript
Data and material availability: Contact authors. Any share should in frame of fair use with acknowledgement of source and/or collaboration.
References
| 1.Khalili H, Derakhshan N, Niakan A, Ghaffarpasand F, Salehi M, Eshraghian H, et al. Effects of oral glibenclamide on brain contusion volume and functional outcome of patients with moderate and severe traumatic brain injuries: A randomized double-blind placebo-controlled clinical trial. World Neurosurg 2017; 101: 130-6. doi: 10.1016/j.wneu.2017.01.103 https://doi.org/10.1016/j.wneu.2017.01.103 PMid:28185976 |
||||
| 2.Maas AIR, Marmarou A, Murray GD, Teasdale SGM, Steyerberg EW. Prognosis and clinical trial design in traumatic brain injury: The IMPACT study. J Neurotrauma 2007; 24: 232-8. https://doi.org/10.1089/neu.2006.0024 PMid:17375987 |
||||
| 3.Guan B, Anderson DB, Chen L, Feng S, Zhou H. Global, regional and national burden of traumatic brain injury and spinal cord injury, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. BMJ Open 2023; 13: e075049. doi: 10.1136/bmjopen-2023-075049 https://doi.org/10.1136/bmjopen-2023-075049 PMid:37802626 PMCid:PMC10565269 |
||||
| 4. Efremov VV, Balyazin-Parfenov IV, Papazova AV, Magomedova EYa, Khantimerian EM. The structure of traumatic brain injury and the availability of specialized neurological care for neurotrauma in Rostov-on-Don during the new SARS-CoV-2 coronavirus infection. South-Russ J Therap Pract 2024; 5: 91-7. Doi: 10.21886/2712-8156-2024-5-2-91-97 https://doi.org/10.21886/2712-8156-2024-5-2-91-97 |
||||
| 5.Maas AIR, Menon DK, Adelson PD, Andelic N, Bell MJ, Belli A, et al. Traumatic brain injury: Integrated approaches to improve prevention, clinical care, and research. Lancet Neurol 2017; 16: 987-1048. https://doi.org/10.1016/S1474-4422(17)30371-X PMid:29122524 |
||||
| 6.Izzy S, Grashow R, Radmanesh F, Chen P, Taylor H, Formisano R, et al. Long-term risk of cardiovascular disease after traumatic brain injury: screening and prevention. Lancet Neurol 2023; 22: 959-70. doi: 10.1016/S1474-4422(23)00241-7 https://doi.org/10.1016/S1474-4422(23)00241-7 PMid:37739576 |
||||
| 7.Lu K, Liang CL, Li PC. Risk factors for myocardial dysfunction after traumatic brain injury: a one-year follow-up study. Injury 2017; 48: 1794-800. https://doi.org/10.1016/j.injury.2017.07.004 PMid:28701282 |
||||
| 8.Boyarinov GA, Deryugina AV, Zaitsev RR, Yakovleva EI, Nikolsky VO, Boyarinova LV, et al. Morphological changes in the myocardium during traumatic brain injury. Forens Medical Exam 2017; 1: 4-7. doi:10.15360/1813-9779-2016-2-20-29 https://doi.org/10.15360/1813-9779-2016-2-20-29 |
||||
| 9.Boyarinov GA, Boyarinova LV, Moshnina EV, Zaitsev PP, Voennov OV, Solovyova OD, et al. Pharmacological correction of hypoxia in patients with combined thoracoabdominal injury. Medial 2014; 1: 23-6. DOI:10.15360/1813-9779-2016-5-42-51 https://doi.org/10.15360/1813-9779-2016-5-42-51 |
||||
| 10.Stein DG, Hoffman SW. Estrogen and progesterone as neuroprotective agents in the treatment of acute brain injuries. Pediatr Rehabil 2003; 6: 13-22. doi: 10.1080/1363849031000095279 https://doi.org/10.1080/1363849031000095279 PMid:12745891 |
||||
| 11.Coppalini G, Salvagno M, Peluso L, Bogossian EG, Quispe Cornejo A, Labbé V, et al. Cardiac injury after traumatic brain injury: clinical consequences and management. Neurocrit Care 2024; 40: 477-85. doi: 10.1007/s12028-023-01777-3 https://doi.org/10.1007/s12028-023-01777-3 PMid:37378852 |
||||
| 12.Ozisik K, Yildirim E, Kaplan S, Solaroglu I, Sargon MF, Kilinc K. Ultrastructural changes of rat cardiac myocytes in a time-dependent manner after traumatic brain injury. Am J Transplant 2004; 4: 900-4. doi: 10.1111/j.1600-6143.2004.00448.x https://doi.org/10.1111/j.1600-6143.2004.00448.x PMid:15147423 |
||||
| 13.Venkata C, Kasal J. Cardiac Dysfunction in adult patients with traumatic brain injury: A prospective cohort study. Clin Med Res 2018; 16: 57-65. doi: 10.3121/cmr.2018.1437 https://doi.org/10.3121/cmr.2018.1437 PMid:30587559 PMCid:PMC6306140 |
||||
| 14.Ivanova GP, Gorobets LN, Litvinov AV, Bulanov VS, Vasilenko LM. A role of progesterone and its metabolites in regulation functions of the brain. SS Korsakov J Neurol Psychiatr 2018; 118: 129‑37. doi: 10.17116/jnevro201811851129 https://doi.org/10.17116/jnevro201811851129 PMid:29927417 |
||||
| 15.Liskova YuV, Stolbova MV, Stadnikov АAA, Salikova SP. Cardioprotective properties of progestines: influence of drospirenone on myocard in experimental heart failure. Russ J Cardiol 2017; 6: 152-7. doi: 10.15829/1560-4071-2017-6-152-15 https://doi.org/10.15829/1560-4071-2017-6-152-157 |
||||
| 16.Coppalini G, Salvagno M, Peluso L, Bogossian EG, Quispe Cornejo A, Labbé V, et al. Cardiac injury after traumatic brain injury: Clinical consequences and management. Neurocrit Care 2024r; 40: 477-85. doi: 10.1007/s12028-023-01777-3 https://doi.org/10.1007/s12028-023-01777-3 PMid:37378852 |
||||
| 17.Dai Q, Likes CE 3rd, Luz AL, Mao L, Yeh JS, Wei Z, et al. A Mitochondrial progesterone receptor increases cardiac beta-oxidation and remodeling. J Endocr Soc 2019; 3: 446-67. doi: 10.1210/js.2018-00219 https://doi.org/10.1210/js.2018-00219 PMid:30746505 PMCid:PMC6364628 |
||||
| 18.Morrissy S, Xu B, Aguilar D, Zhang J, Chen QM. Inhibition of apoptosis by progesterone in cardiomyocytes. Aging Cell. 2010 Oct;9(5):799-809. doi: 10.1111/j.1474-9726.2010.00619.x. PMID: 20726854; PMCID: PMC4133411. https://doi.org/10.1111/j.1474-9726.2010.00619.x PMid:20726854 PMCid:PMC4133411 |
||||
| 19.Lan C, Cao N, Chen C, Qu S, Fan C, Luo H, et al. Progesterone, via yes-associated protein, promotes cardiomyocyte proliferation and cardiac repair. Cell Prolif 2020; 53: e12910. doi: 10.1111/cpr.12910 https://doi.org/10.1111/cpr.12910 PMid:33047378 PMCid:PMC7653240 |
||||
| 20.Amirkhosravi L, Khaksari M, Amiresmaili S, Sanjari M, Khorasani P, Hashemian M. Evaluating the neuroprotective effects of progesterone receptors on experimental traumatic brain injury: The PI3K/Akt pathway. Brain Behav 2023; 13) :e3244. doi: 10.1002/brb3.3244 https://doi.org/10.1002/brb3.3244 PMid:37661235 PMCid:PMC10636406 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER