Impact of smoking on left and right ventricular systolic function and left ventricular mass in young healthy males: A two- and three-dimensional echocardiographic study
ORIGINAL RESEARCH ARTICLE
Impact of smoking on left and right ventricular systolic function and left ventricular mass in young healthy males: A two- and three-dimensional echocardiographic study
Article Summary
- DOI: 10.24969/hvt.2025.611
- CARDIOVASCULAR DISEASES
- Published: 30/11/2025
- Received: 19/09/2025
- Revised: 08/11/2025
- Accepted: 08/11/2025
- Views: 1568
- Downloads: 1409
- Keywords: Echocardiography, young healthy males, smoking, left ventricle, left ventricular mass, right ventricle
Address for Correspondence: Muhammad Aly Mahmoud Fayed, Department of Cardiovascular Medicine, Kasr Alainy Faculty of Medicine, Cairo University, Cairo, Egypt
Email: Dr.muhammadfayed85@kasralainy.edu.eg
ORCID: Muhammad Fayed - 0009-0004-3187-3215; Amr Elfaramawy - 0000-0003-1756-9132; Azza Farrag - 0000-0002-8901-4673; Heba Eldeeb - 0000-0002-1320-2600
Facebook: Muhammad Fayed - https://facebook.com/mohamed.a.fayed.5
Impact of smoking on left and right ventricular systolic function and left ventricular mass in young healthy males: A two- and three-dimensional echocardiographic study
Muhammad Fayed, Amr Elfaramawy, Azza Farrag, Heba Eldeeb
Department of Cardiovascular Medicine, Kasr Alainy Faculty of Medicine, Cairo University, Cairo, Egypt
Abstract
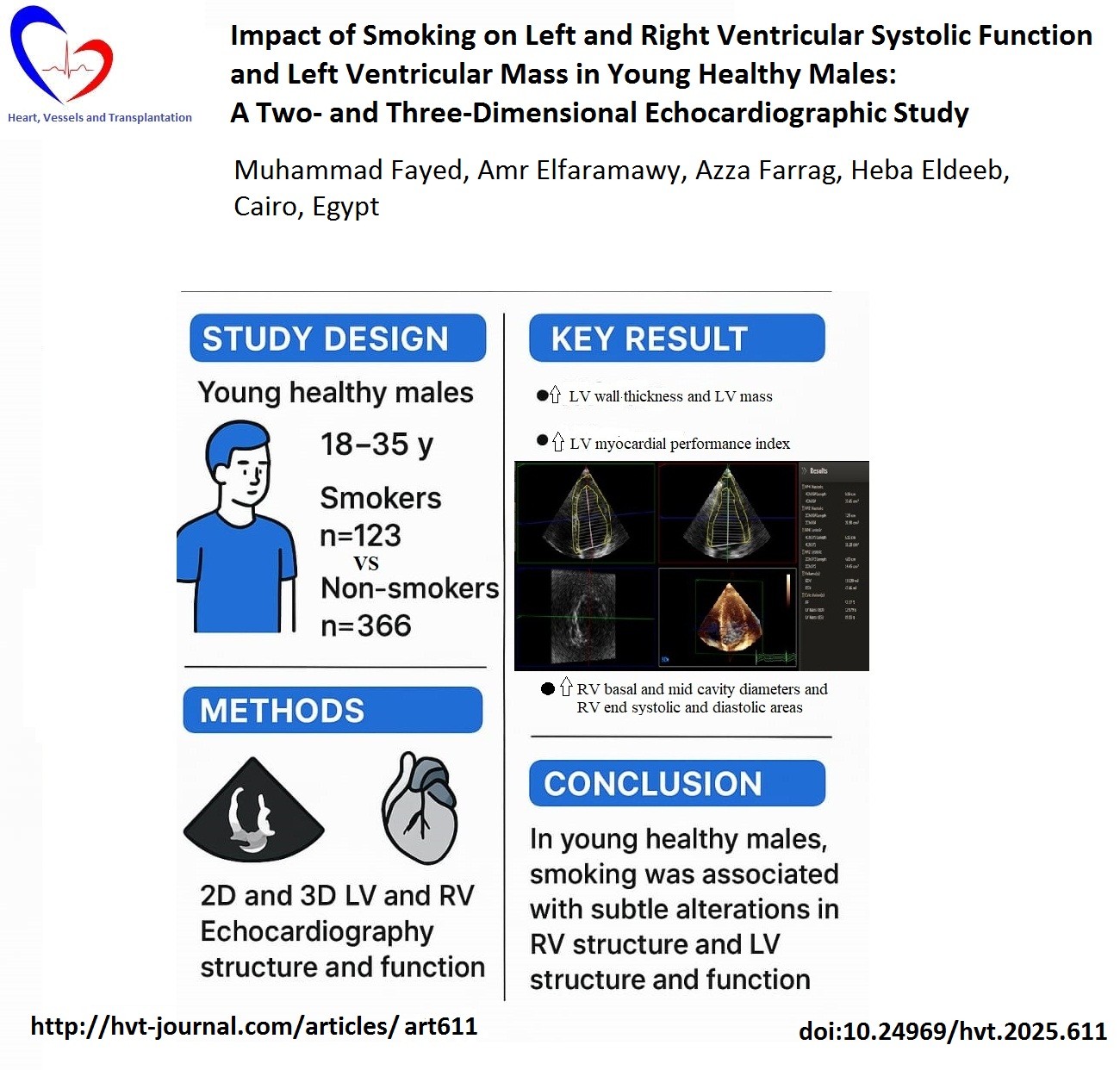
Objective: Cigarette smoking has been associated with increased mortality, mainly due to cardiovascular diseases (CVDs). The relationship between tobacco use and CVDs is complex, and that between cigarette smoking and echocardiographic measurements of right ventricular (RV) and left ventricular (LV) structure and function has been poorly studied. This study aimed to evaluate the effect of smoking on LV and RV systolic functions and LV mass (LVM) in young healthy males using two (2D) and three-dimensional (3D) transthoracic echocardiography (TTE).
Methods: Data for this observational study were collected by screening of 1013 males (smokers and non-smokers) including relatives of patients, workers, medical students and hospital clerks in Cairo University hospitals for thirty months and after history taking and clinical examination, exclusion was done for any candidate with any comorbidities or history of any drug addiction or cannabis consumption or athletes and/or those who perform forcible sports, any candidate with poor echogenic window or any valvular lesions. Overall, 489 healthy males aged 18–35 years were grouped into smoker (123 participants) and non-smoker (366 participants) groups, then subjected to standard 2D TTE after exclusion of to evaluate dimensions and systolic function of LV and RV, then 3D TTE was used to evaluate LV volumes, systolic function and LVM.
Results: Among the smoker group, the 2D TTE parameters revealed a trend towards increasing most of the values. However, only septal and posterior walls thickness (p=0.03 and p=0.01, respectively), LV myocardial performance index (p = 0.029), LVM and that indexed to body surface are (BSA) (p=0.004 and p=0.008 respectively), RV basal and mid cavity diameters (both p<0.0001), RV end-diastolic area and that indexed to BSA (both p<0.0001), and end systolic area and that indexed to BSA (p=0.016 and p=0.041, respectively) showed statistically significant higher values. The LV 3D TTE parameters also revealed similar trend in the smoker group. However, only LVM and that indexed to BSA (both p<0.0001) showed statistically significant higher values.
Conclusion: In young healthy males, smoking was associated with subtle alterations in RV structure and LV structure and function.
Key words: Echocardiography, young healthy males, smoking, left ventricle, left ventricular mass, right ventricle
Introduction
Cigarettes are the most commonly used tobacco product, and the World Health Organization (WHO) classifies smoking addiction as an epidemic (1). Cigarette smoking significantly increases mortality, primarily due to its role in cardiovascular diseases (CVDs) and cancer development (2). Regardless of conventional risk factors like hypertension, dyslipidemia, or diabetes, smoking is recognized as an independent risk factor for CVDs, contributing to cardiovascular damage through mechanisms separate from traditional risk factors (3).![]()

Graphical abstract
Smoking is one of the most significant preventable risk factors for atherosclerosis (4). Beyond its role in atherosclerosis, smoking has been linked to structural changes in the heart, including increased left ventricular (LV) mass (LVM) and abnormal LV geometry compared with non-smokers, even in the absence of major cardiovascular events (5). Studies examining the relationship between smoking and cardiac structure and function in community-based populations have produced conflicting results. While some research has found an association between active smoking and increased LVM (6-10), others have reported neutral (11) or even inverse relationships (12). In addition, the relationship between smoking and LV and right ventricular (RV) systolic functions remains controversial (7, 9, 10, 13-16). LVM is a known risk factor for cardiovascular morbidity and mortality (17-19). Despite its prognostic importance, its measurement and clinical implications have not been firmly established (20). Additionally, while factors contributing to increased LVM have been extensively studied in older (21, 22) and diseased populations (23), research on young, healthy individuals remains limited. Most studies in this group have involved small sample sizes (24, 25), leading to reduced statistical power and inconclusive findings.
Echocardiography and cardiac magnetic resonance (cMRI) are the two most commonly used imaging methods for the assessment of LVM. Although cMRI is considered the gold standard for assessing LVM, echocardiography is a well validated, non-invasive method that is more widely used in clinical practice (26). However, traditional two-dimensional (2D) transthoracic echocardiography (TTE) has limitations, as it relies on geometric assumptions of uniform chamber size and shape. Additionally, the unintended use of oblique planes can lead to an overestimation of LVM measurements in M-mode, while foreshortening in apical views can result in underestimation (27). To overcome these limitations, three-dimensional (3D) TTE provides more accurate LVM and volume measurements, with reproducibility comparable to that of cMRI (28).
The primary aim of this study was to evaluate the effect of smoking on LV and RV systolic function and LVM in young healthy males using 2D- and 3D-TTE. The secondary aim was to compare measuring LVM by M-mode echocardiography (linear method) and three-dimensional echocardiography in these apparently healthy males.
Methods
Study design and population
This study is an observational study, which was conducted in Cario University, Cairo, Egypt hospitals for thirty months. Screening was done for 1013 males (smokers and non-smokers) including relatives of patients, workers, medical students and hospital clerks (Fig. 1) but after exclusion of candidates older than 35 years or less than 18 years (at time of the study), only 758 candidates were subjected to history taking and clinical examination. After exclusion of any candidate with hypertension, diabetes or any other comorbidities or history of any drug addiction or cannabis consumption or athletes and/or those who perform forcible sports, only 608 candidates were subjected to TTE study. Sixty-nine candidates were found to be technically difficult, had mild valvular lesions or had poor echocardiographic window, so they were excluded. The remaining 539 candidates were grouped into two groups (smoker group and non-smoker group) and after matching the two groups according to age, body surface area (BSA) using the formula of Du Bois (29) and body mass index (BMI), fifty candidates were excluded. Therefore, our study population included 489 candidates: of whom 123 candidates (25.2 %) represented the smoker group (Fig. 1) and rest 366 – non-smoker group.
Informed consent was obtained from all participants in our study. The study was performed according to Helsinki 2024 convention on human studies and the protocol was approved by Ethics Committee of Cairo University, Cairo, Egypt.
Baseline variables
All participants underwent history taking and clinical examination with assessment of age, BSA, BMI, history of drug abuse, participation in competitive sports, and comorbidities, structural heart disease.
Smoking assessment
The history of smoking was taken by a questionnaire to measure the average daily consumption of cigarettes [cigarette per day (CPD)] and time to first cigarette of the day (TTFC). CPD is given codes as zero if CPD equals 1-10; one if CPD equals 11-20; two if CPD equals 21-30; three if CPD greater than or equals 31. TTFC is given codes zero if TTFC greater than or equals 61 min; one if TTFC equals 31-60 min; two if TTFC equals 6-30 min; three if TTFC lesser than or equals 5 min. By summing the two previous scores, a scale with a range of 0-6 will be given which will measure the heaviness of smoking index (HIS) (30).
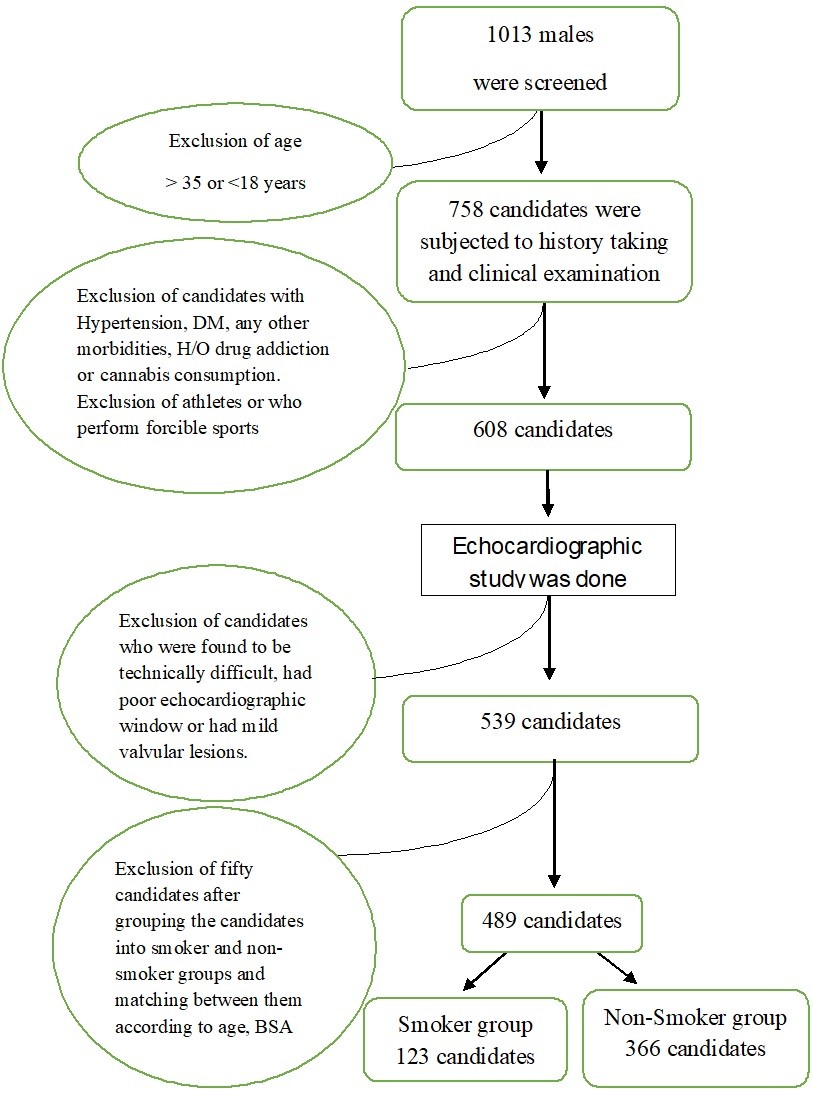
Figure 1. Flowchart showing participants recruitment and number of candidates studied
Echocardiographic study
2D transthoracic echocardiography (TTE)
It was performed using Philips EPIQ 7 unit (Philips Medical Systems, Andover, MA, USA) equipped with 2.5 MHz X5-1 transducer. The patients were studied in the left lateral decubitus position. Image and Doppler acquisitions were obtained at held end-expiration. Standard views for TTE were parasternal long-axis and short-axis views, and apical two-, three-, four-, and five-chamber views.
Quantification of the cardiac chambers was done as per the American Society of Echocardiography and the European Association of cardiovascular imaging for assessment of cardiac chambers (31) including measurement of aortic root diameter at sinus of Valsalva and left atrium anteroposterior diameter by 2D-guided linear measurements. M-Mode guided linear measurements of LV end-diastolic (LVEDD) and end-systolic diameters (LVESD), interventricular septal (IVST) and posterior walls thickness (PWT) at end diastole, and LV ejection fraction (LVEF) were done.
Relative wall thickness (RWT) was calculated using this formula, RWT = 2 × PWT/LVEDD, while LVM was calculated by M-Mode linear method using the Devereux formula: LVM = 0.8x{1.04[(IVST + LVEDD + PWT)3 - LVEDD3]}+ 0.6. All measurements were performed at end-diastole (31, 32)
Myocardial performance index (Tei Index, MPI): The pulsed Doppler probe was placed between mitral inflow and LV outflow. Doppler time intervals were measured from mitral inflow and LV outflow Doppler tracings, as described by Tei and co-workers (33). The interval ‘a’ from end to onset of mitral inflow is equal to the sum of isovolumic contraction time (ICT), ejection time (ET), and isovolumic relaxation time (IRT). ET ‘b’ is derived from the duration of the LV outflow Doppler velocity profile. The sum of ICT and IRT was obtained by subtracting b from a. The MPI was calculated from the following formula: (a - b)/b.
RV linear dimensions (basal, mid and longitudinal RV) were measured at end-diastole, RV end-diastolic area (RV EDA) and end systolic area (RV ESA) were measured by manual tracing of RV endocardial border at end-diastole and at end-systole (31).
Estimation of RV contractile function by tricuspid annular plane systolic excursion (TAPSE) measuring tricuspid annular longitudinal excursion by M-mode between end-diastole and peak systole. Fractional area change (FAC) of RV measured in RV-focused apical four-chamber view: RV FAC (%) = (RV EDA- RV ESA)/RV EDA X 100 (31).
3D transthoracic echocardiography (TTE)
It was performed using Philips EPIQ 7 unit (Philips Medical Systems, Andover, MA, USA) equipped with a dedicated broadband, wide angle, matrix array transducer. Images were acquired from the apical window, with the patient in the left lateral decubitus position. Care was taken to include the entire LV cavity within the pyramidal scan volume. 3D echo datasets were acquired and obtained over 4 different electrocardiogram gated cardiac cycles during a short breath hold to avoid stitching artifacts.
Measurements of LV end-diastolic (LV EDV) and the end-systolic (LV ESV) volumes, LVEEF and LVM were performed off-line. The 3D echo datasets were analyzed with a prototype version of 3D QLab 10 software (cardiac motion quantification (CMQ); Philips Medical Systems) that allowed for semi-automatic detection of endocardial and epicardial borders. The 3D volume datasets were displayed in 3 different cross-sections that can be modified interactively. Markers were then placed onto the mitral annulus and the apex, both in end-diastole and end-systole. Using these markers, the software program created truncated ellipsoid end-diastolic and end-systolic 3D meshes of the LV. The end-diastolic and the end-systolic 3D meshes deformed until equilibrium were reached between closeness to the borders and minimal curvature. If needed, manual adjustments were applied. The volumes of the obtained 3D-end-diastolic and end-systolic LV meshes included the papillary muscles into the LV cavity and corresponded to the LV EDV and LV ESV and so LV EF was calculated (31, 34, 35).
Measuring of LVM was done by detecting the epicardial contours, manual corrections were then applied, if needed. These epicardial contours were used to calculate the myocardial volume by subtracting the end-diastolic endocardial volume from the end-diastolic epicardial volume. The difference between these two volumes was then multiplied by the specific mass of myocardial tissue (1.05 g/ml) to obtain LVM (36, 37) as shown in Figure 2.
Statistical analysis
Data was statistically analyzed using the Statistical Package of Social Science Software program (SPSS), version 20 (IBM, New York, USA).
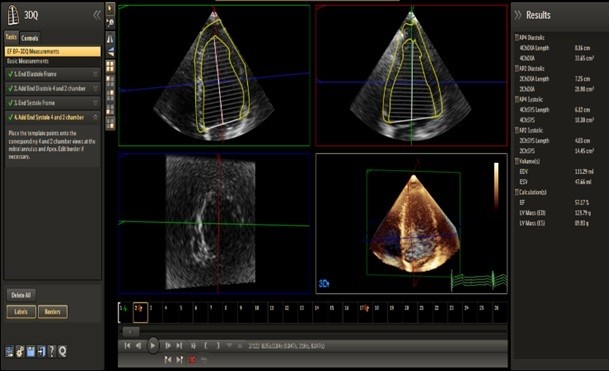
Figure 2. Three-D measurement of LV mass of one of cases
D-dimensional, LV - left ventricular
Continuous variables were described in terms of mean (standard deviation, SD) while categorical variables were described in terms of frequencies (number of cases) and percentages (%) when appropriate.
Mann Whitney U test was used for independent samples for comparison between smoker and non-smoker groups with continuous variables, which were not normally distributed. The confidence interval was set to 95% and the margin of error was set to 5%. The p value was considered significant if <0.05 and highly significant if < 0.01. Wilcoxon Signed Ranks test was used to compare between two different modalities: 3D TTE and M-mode linear method, which were used to assess LVM. Bland Altman analysis was used to evaluate the differences between the M-mode linear method and 3D TTE as regard to assessment of LVM.
Results
The study population included 489 candidates: of whom 123 candidates (25.2%) represented the smoker group. Baseline clinical characteristics of the whole study population showed that the mean age was 26.3 (4.8) years, BSA was 1.97 (0.15) m2 and BMI was 25.34 (3.91) kg/m2 as in Table 1.
|
Table1. Study population baseline characteristics |
|
|
Variables |
mean (SD) |
|
Age, years |
26.3 (4.8) |
|
Body surface area, m2 |
1.97 (0.15) |
|
Body mass index, kg/m2 |
25.34 (3.91) |
Smoking parameters of the smoking group showed that the average years of smoking were 5.0 (3.4) years, the mean amount of cigarette smoked per day were 16 (9) cigarettes/day, the mean TTFC was 32.0 (21.6) min and the mean HIS score was 2.4 (1.3) as shown in Table 2. There were no significant correlations between these smoking parameters and the 2D or 3D TTE parameters.
|
Table 2. Smoking parameters of the smoking group |
|
|
Variables |
mean (SD) |
|
Average years of smoking, years |
5.0 (3.4) |
|
Amount of cigarette smoked per day, cigarettes/day |
16 (9) |
|
Time to first cigarette of the day (TTFC), min |
32.0 (21.6) |
|
Heaviness of smoking index (HIS) score |
2.4 (1.3) |
Effect of smoking on 2D TTE variables
The comparison of LV 2D TTE parameters between smoker and non-smoker groups demonstrated that there was a trend towards increased values of most of the LV parameters in the smoker group. However, septal wall thickness IVST (p=0.03), PWT (p=0.01), LV MPI (p=0.029), LVM (p=0.004) and LVM indexed to BSA (LVMI) (p=0.008) showed statistically significant higher values in smokers as compared to non-smokers.
Regarding RV 2D TTE parameters, the comparison displayed also a trend towards increased values of most of the parameters in the smoker group. However, RV basal diameter (p<0.0001], RV mid diameter (p<0.0001), RVEDA (p<0.0001) and RVEDA indexed to BSA (p<0.0001), RVESA (p=0..016] and RVESA indexed to BSA (p=0.041( showed statistically significant higher values in smokers group as compared to non-smokers group, as shown in Table 3.
|
Table 3. Comparison of 2D echocardiographic parameters of the left ventricle, left atrium and aorta and RV ventricle between smoker and non-smoker groups |
|||
|
Variables |
Smoker (n=123) |
Non -smoker (n=366) |
p* |
|
LVEDD, cm |
4.91 (0.47) |
4.85 (0.45) |
0.198 |
|
LVEDD indexed to BSA, cm/m2 |
2.5 (0.29) |
2.48 (0.28) |
0.585 |
|
LVEDV, ml |
114.67(24.67) |
111.66 (23.87) |
0.211 |
|
LVEDV indexed to BSA, ml/m2 |
58.21 (12.53) |
57.05 (12.48) |
0.341 |
|
LVESD, cm |
3.22 (0.32) |
3.16 (0.35) |
0.182 |
|
LVESD indexed to BSA, cm/m2 |
1.64 (0.21) |
1.62 (0.22) |
0.436 |
|
LVESV, ml |
41.95 (9.98) |
40.68 (10.91) |
0.231 |
|
LVESV indexed to BSA, ml/m2 |
21.34 (5.23) |
20.82 (5.81) |
0.277 |
|
IVST, cm |
0.85 (0.1) |
0.82 (0.11) |
0.003 |
|
PWT, cm |
0.81 (0.1) |
0.78 (0.1) |
0.01 |
|
LV EF, % |
63.16 (5.24) |
63.58 (5.55) |
0.56 |
|
LV MPI |
0.39 (0.04) |
0.38 (0.04) |
0.029 |
|
RWT |
0.33 (0.05) |
0.33 (0.05) |
0.202 |
|
LVM, gr |
139.58 (30.95) |
129.99 (26.4) |
0.004 |
|
LVMI, gr/m2 |
70.71 (15.06) |
66.37 (13.65) |
0.008 |
|
LA diameter, cm |
3.35 (0.38) |
3.36 (0.4) |
0.904 |
|
LA diameter indexed to BSA, cm/ m2 |
1.71 (0.23) |
1.72 (0.23) |
0.881 |
|
Aortic root diameter at sinus of Valsalva, cm |
2.92 (0.31) |
2.91 (0.29) |
0.967 |
|
Aortic root diameter indexed to BSA, cm/ m2 |
1.49 (0.21) |
1.49 (0.18) |
0.534 |
|
RV basal, cm |
3.62 (0.44) |
3.39 (0.44) |
<0.0001 |
|
RV mid, cm |
3.04 (0.44) |
2.79 (0.51) |
<0.0001 |
|
RV longitudinal, cm |
7.2 (0.76) |
7.08 (0.73) |
0.15 |
|
RVEDA, cm2 |
18.41 (2.68) |
17.12 (2.45) |
<0.0001 |
|
RVEDA indexed to BSA, cm2 |
9.36 (1.48) |
8.76 (1.39) |
<0.0001 |
|
RVESA, cm2 |
9.68 (1.98) |
9.18 (1.82) |
0.016 |
|
RVESA indexed to BSA, cm2 |
4.92 (1.04) |
4.7 (1.0) |
0.041 |
|
FAC, % |
47.53 (6.64) |
46.45 (6.77) |
0.091 |
|
TAPSE, cm |
2.37 (0.34) |
2.35 (0.33) |
0.587 |
|
*p value is significant < 0.05 and is highly significant < 0.01, Data are presented as mean (SD), Mann Whitney U test BSA - body surface area, FAC - Fractional area change, LA - Left atrium, LV - Left ventricle, LV EF - Left ventricle ejection fraction, LV MPI - Left ventricle myocardial performance index, LVEDD - left ventricular end-diastolic dimension, LVEDV - left ventricular end-diastolic volume, LVESD - left ventricular end-systolic dimension, LVESV - left ventricular end-systolic volume, PWT - posterior wall thickness, RV - right ventricle, RVEDA - right ventricle end-diastolic area, RVESA - right ventricle end-systolic area, RWT - relative wall thickness, IVST - interventricular septum thickness, TAPSE - tricuspid annular plane systolic excursion |
|||
Smoking and 3D TTE variables
Comparison of LV 3D TTE parameters between smoker and non-smoker groups, revealed no differences in LV volumes and LVEF, however, only LVM (p<0.0001) and LVMI (p<0.0001] showed statistically significant higher values, as shown in Table 4.
|
Table 4. Comparison between smoker and non-smoker groups regarding 3D echocardiographic parameters of the left ventricle |
|||
|
Variables |
Smoker (n=123) |
Non -smoker (n=366) |
P* |
|
LVEDV, ml |
123.76 (22.91) |
120.23 (22.98 |
0.158 |
|
LVEDV indexed to BSA, ml/ m2 |
62.9 (11.99) |
61.44 (12.15) |
0.268 |
|
LVESV, ml |
47.4 (9.03) |
46.12 (10.37) |
0.304 |
|
LVESV indexed to BSA, ml/ m2 |
24.09 (4.68) |
23.59 (5.53) |
0.354 |
|
LV EF, % |
61.48 (4.17) |
61.58 (4.73) |
0.879 |
|
LVM, gr |
154.23 (25.89) |
142.59 (23.2) |
<0.0001 |
|
LVMI, gr/m2 |
78.19 (12.5) |
72.8 (12.13) |
<0.0001 |
|
*p value is highly significant < 0.01, Data are presented as mean (SD), Mann Whitney U test BSA - body surface area, LV - Left ventricle, LV EF - left ventricular ejection fraction, LVEDV - left ventricular end-diastolic volume, LVESV - left ventricular end-systolic volume, LVM – left ventricular mass, LVMI – LVM indexed to BSA |
|||
Comparison of LVM measured by M-mode echocardiography (linear method) and 3D TTE
In the whole study population (489 candidates), LVM measured by 3D TTE was significantly higher (p<0.0001) when compared to that measured by the linear method [145.52 (24.41) gr vs. 132.4 (27.89) gr, respectively] and [LVMI 74.15 (12.44) gr/ m2 and 67.46 (14.13) gr/m2, respectively].
Wilcoxon Signed Ranks test revealed that LVM and LVMI values measured by 3D TTE were higher than that measured by the linear method in 455 candidates (positive ranks) and were higher in the linear method compared with 3D TTE in only 34 candidates (negative ranks). Bland-Altman analysis in the whole study population showed a trend toward lower values of LVM measured by M-mode compared with that measured by 3D TTE by demonstrating a bias of -13.12 gr with 95% limits of agreement at ± 10.86 gr, the upper limit of agreement = 8.16 gr and the lower limit of agreement = -34.39 gr (Fig. 3).
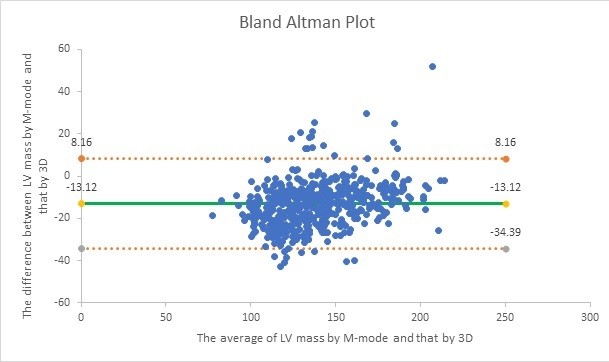
Figure. 3 Bland Altman analysis of agreement between LVM measured by M-mode and that measured by 3D TTE
LV – left ventricular, LVM – left ventricular mass, 3D TTE – 3-dimensional transthoracic echocardiography
Also, Bland-Altman analysis showed a trend toward lower values of LVMI measured by M-mode compared with that measured by 3D TTE by demonstrating a bias of -6.7 gr/m2 with 95% limits of agreement at ± 5.58 gr/m2, the upper limit of agreement = 4.25 gr/m2 and the lower limit of agreement = -17.64 gr/m2 (Fig. 4).
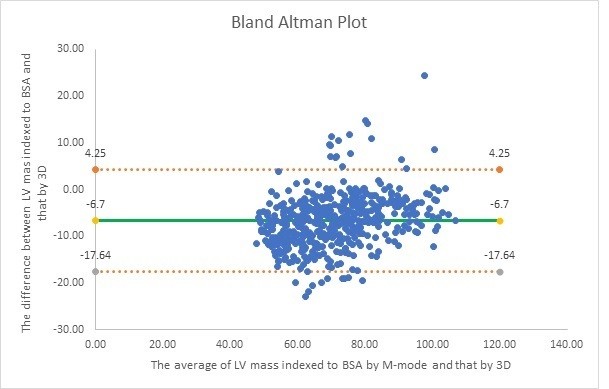
Figure 4. Bland-Altman analysis of agreement between LVMI measured by M-mode and that measured by 3D TTE
LV – left ventricular, LVMI – left ventricular mass index, 3D TTE – 3-dimensional transthoracic echocardiography
Discussion
Smoking has been associated with structural changes in the heart including LVM and LV geometry, 5 these changes in myocardial structure and function can be evaluated by echocardiography.
TTE is a widely used imaging technique known for its safety, absence of ionizing radiation, affordability, portability, and quick to perform. It remains the most commonly employed method worldwide for assessing cardiac chamber dimensions and LV wall thickness (38). This study was conducted on young healthy males aiming to evaluate the effect of smoking on LVand RV structure and systolic function and LVM using TTE and it was found that the 2D TTE parameters revealed a trend towards increasing most of the values. However, only IVST and PWT, LV MPI, LVM and LVMI, RV basal and mid cavity diameters, RV EDA and that indexed to BSA, and RV ESA and that indexed to BSA showed statistically significant higher values. The LV 3D TTE parameters also revealed that only LVM and LVMI showed statistically significant higher values in smokers as compared with non-smokers.
Previous 2D TTE studies have reported conflicting findings regarding the impact of smoking on structure and function of LV and RV compared with our data (7, 9, 10, 12).
The ECHO-SOL study (10) (Echocardiographic Study of Hispanics/ Latinos) examined the effect of smoking in 1818 of Hispanic/Latino population of the United States with mean age 56.4 years. In agreement to the current study, comparing the current smokers with never smokers population showed significant increase in LVEDV and LVESV, and increased RVEDA and LVM, which tend to be statistically significant (p=0.06 and p=0.09, respectively). But in contrast to our data, the ECHO-SOL showed significant decrease of LVEF and RV FAC. Also, the current study showed a significant increase of LV MPI in smoker group which may be considered as a subtle LV systolic and diastolic dysfunction. The decreased LVEF and RV FAC in smoker population of ECHO-SOL can be explained by the differences of mean age of both studies. The ECHO-SOL study population was significantly older than the current population by an average of 30 years, so ECHO-SOL study population was exposed to more years of smoking and more cigarettes smoked and altered RV function was associated with more cigarettes smoked.
Cigarette smoking is also the most common risk factor for development of chronic obstructive pulmonary disease (COPD) (39) which was not an exclusion criterion in ECHO-SOL. COPD is associated with pulmonary hypertension (40) and RV enlargement, LV enlargement, as well as LV systolic and diastolic dysfunction (41). Even subclinical impairment of lung function has been shown to be associated with LV systolic and diastolic dysfunction (42), which could potentially contribute to RV dysfunction as well.
The ARIC Study (9) (Atherosclerosis Risk in Communities) examined the effect of smoking in 4580 participants of the United States free of overt coronary artery disease, heart failure, and significant valvular disease with mean age 74 years. Comparing their current smokers with never smokers population showed statistically significant increase of IVST,, LVM and LVMI, aligning with our findings. In contrast to our data, there is a marginally higher LVEF in current smokers. Also, ARIC Study showed no significant association between RV structural and functional features and smoking status. This increase in LVEF may be influenced by cardiac geometry (43) and was known to increase in parallel with LVM/volume ratio (44). In addition ARIC study researchers found that the difference in LVEF between current and never smokers became no longer apparent after adjusting for LVM/volume ratio in multivariable analysis, suggesting that the higher LVEF seen in current smokers could be more a consequence of variation in LV geometry rather than resulting from enhanced LV systolic function. Furthermore, in the ARIC Study, the global longitudinal LV strain, which has been reported as a more sensitive measure of systolic function, (45) did not differ among their study groups.
Regarding 3D TTE parameters, the comparison between the smokers and non-smokers groups in the current study showed statistically significant differences only for LVM and LVMI. 3D TTE was chosen in the current study to detect the effect of smoking on LV systolic function and structure especially the LVM.
However, previous studies as Multi-Ethnic Study of Atherosclerosis (MESA) (7) and LARGE Heart study (12) used cMRI as a method of assessment to detect the effect of smoking. The MESA study (7) examined 4869 participants from 4 racial/ethnic groups: African, American, Hispanic, and Asian enrolled from six U.S. communities with mean age 62 years to assess the effect of traditional cardiovascular risk factors including smoking on LV mass, volume, and systolic function by cMRI. Current smokers of MESA study were associated with higher LVM in agreement to our data but lower LVEF in contrast to our data, this may be explained by the larger sample size in MESA study. Also the use of cMRI, as a method of assessment in MESA study, permitted researchers to identify relationships of smoking with subclinical alterations in LV size and systolic function that were difficult to be detected using previously available noninvasive methods as echocardiography, so MESA study allowed the detection of very small differences in LV mass, volumes, and systolic function in relation to smoking.
The LARGE Heart study (12) also used cMRI as a method of assessment of LVM and its association with lifestyle including cigarette smoking. But in comparison to MESA, they examined healthy Caucasian male army recruits from United Kingdom which were younger study population (with mean age 19.8 years) and a smaller sample size (n = 541). Unexpectedly in LARGE Heart study, there was an inverse relationship, with LVM being less in current smokers. Researchers tried to suggest that such an association was mediated through blood pressure that the systolic blood pressure (SBP) was significantly lower in current smokers. However, such an observation was misleading. Although cross-sectional studies demonstrate that blood pressure is often lower amongst smokers (46), brachial artery SBP is affected by pulse pressure amplification (47) — a phenomenon that exaggerates aortic pulse pressure when measured peripherally. This effect is particularly pronounced in young individuals and is known to be diminished by smoking. As a result, reduced pulse pressure amplification in smokers could lead to lower brachial artery SBP, especially in studies involving young populations. Moreover, aortic SBP, a key determinant of LV workload, has been found to be higher in young smokers when measured using applanation tonometry (47). This suggests that the observed lower LVM in smokers within the LARGE Heart study cannot be solely explained by reduced SBP, and an alternative explanation should be explored.
Comparison between measuring LVM by M-mode linear method and 3D echocardiography
Our data showed that the LVM and LVMI measured by 3D TTE was higher than that measured by the M-mode linear method. Data from previous studies showed that M-mode linear method measuring LVM (probably due to oblique cuts) usually overestimated LVM in comparison to cMRI, which is the gold standard for assessment of LVM..
The LVM measurement by 3D TTE showed improved accuracy compared to conventional echo techniques. In the study done by Yap et al. (48) which evaluated the accuracy of real-time 3D TTE in determining LVM compared to cMRI, their data showed good agreement between the two modalities.
Meta-analysis done by Shimada and Shiota (49) detected the accuracy of LVM measurement by 3D TTE and evaluated the controversial data on the systematic bias in LVM assessment by 3D TTE, they revealed the existence of significant underestimation in the past, improvement of accuracy over the past decade, and excellent accuracy with most recent 3D TTE technologies. Inaccuracy of LVM measurement can result mainly from 2 sources: tracking of the endocardium and the epicardium. Tracing the inner side of the endocardium leads to underestimation of cardiac volumes and overestimation of LVM, which has been repeatedly shown in previous series of studies (50). In contrast, tracking the inner side of the epicardium leads to the underestimation of LVM. They found that the underestimation arising from epicardial tracking had outweighed the overestimation from endocardial tracking, resulting in a systematic underestimation of LVM in the earlier studies.
Study limitations
Firstly, we described results of a single-center study with a relatively small number of the studied population and included males only so other studies including larger number with females are needed. Secondly, estimation of tissue Doppler measurements and diastolic function assessment were not performed to detect the effect of smoking on diastolic function. Thirdly, our study did not include speckle-tracking echocardiography to detect LV and RV global longitudinal strain which has been reported as a more sensitive measure of the effect of smoking on systolic function and can detect even any subtle changes in systolic function.
Conclusion
In young healthy males, smoking was associated with increased wall thickness and LVM together with increased LV MPI. Also, smoking was associated with increased RV basal and mid-cavity diameters in addition to increased RV end systolic and diastolic areas. These findings suggest that smoking was associated with subtle alterations in RV structure and LV structure and function.
Ethics: Informed consent was obtained from all participants in our study. The study was performed according to Helsinki 2024 convention on human studies. The study protocol was approved by Ethics Committee of Faculty of Medicine, Cairo University, Cairo, Egypt.
Peer-review: External and internal
Conflict of interest: None to declare
Authorship: M.F., A.E., A.F., H.E. equally contributed to the study and manuscript preparation , approved the final manuscript and
fulfilled the authorship criteria
Acknowledgements and Funding: None to declare
Statement on A.I.-assisted technologies use: Authors used A.I. (CHAT GPT) in suggestion of design of graphical abstract
Data and material availability: Contact authors. Any share should in frame of fair use with acknowledgement of source and/or collaboration.
References
| 1. World Health Organization. WHO Report on the Global Tobacco Epidemic, 2009: implementing smoke-free environments: executive summary (No. WHO/NMH/TFI/09.1). World Health Organization 2009. | ||||
| 2. West R. Tobacco smoking: Health impact, prevalence, correlates and interventions. Psychol Health 2017; 32: 1018-36. https://doi.org/10.1080/08870446.2017.1325890 PMid:28553727 PMCid:PMC5490618 |
||||
| 3. Conklin DJ, Schick S, Blaha MJ, Carll A, DeFilippis A, Ganz P, et al. Cardiovascular injury induced by tobacco products: assessment of risk factors and biomarkers of harm. A Tobacco Centers of Regulatory Science compilation. Am J Physiol Heart Circ Physiol 2019; 316: H801-27. https://doi.org/10.1152/ajpheart.00591.2018 PMid:30707616 PMCid:PMC6483019 |
||||
| 4. Messner B, Bernhard D. Smoking and cardiovascular disease: mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler Thromb Vasc Biol 2014; 34: 509-15. https://doi.org/10.1161/ATVBAHA.113.300156 PMid:24554606 |
||||
| 5. Gidding SS, Liu K, Colangelo LA, Cook NL, Goff DC, Glasser SP, et al. Longitudinal determinants of left ventricular mass and geometry: the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Circ Cardiovasc Imaging 2013; 6: 769-75. https://doi.org/10.1161/CIRCIMAGING.112.000450 PMid:23922005 PMCid:PMC3873157 |
||||
| 6. Gardin JM, Arnold A, Gottdiener JS, Wong ND, Fried LP, Klopfenstein HS, et al. Left ventricular mass in the elderly: the Cardiovascular Health Study. Hypertens 1997; 29: 1095-103. https://doi.org/10.1161/01.HYP.29.5.1095 PMid:9149672 |
||||
| 7. Heckbert SR, Post W, Pearson GD, Arnett DK, Gomes AS, Jerosch-Herold M, et al. Traditional cardiovascular risk factors in relation to left ventricular mass, volume, and systolic function by cardiac magnetic resonance imaging: the Multiethnic Study of Atherosclerosis. J Am Coll Cardiol 2006; 48: 2285-92. https://doi.org/10.1016/j.jacc.2006.03.072 PMid:17161261 PMCid:PMC1794681 |
||||
| 8. Markus MR, Stritzke J, Baumeister SE, Siewert U, Baulmann J, Hannemann A, et al. Effects of smoking on arterial distensibility, central aortic pressures and left ventricular mass. Int J Cardiol 2013; 168: 2593-601. https://doi.org/10.1016/j.ijcard.2013.03.045 PMid:23597572 |
||||
| 9. Nadruz Jr W, Claggett B, Gonçalves A, Querejeta-Roca G, Fernandes-Silva MM, Shah AM, et al. Smoking and cardiac structure and function in the elderly: the ARIC Study (Atherosclerosis Risk in Communities). Circ Cardiovasc Imaging 2016; 9: e004950. https://doi.org/10.1161/CIRCIMAGING.116.004950 PMid:27625349 PMCid:PMC5193104 |
||||
| 10. Leigh JA, Kaplan RC, Swett K, Balfour P, Kansal MM, Talavera GA, et al. Smoking intensity and duration is associated with cardiac structure and function: the ECHOcardiographic Study of Hispanics/Latinos. Open Heart 2017; 4: e000614. https://doi.org/10.1136/openhrt-2017-000614 PMid:28761681 PMCid:PMC5515129 |
||||
| 11. Hasegawa T, Boden-Albala B, Eguchi K, Jin Z, Sacco RL, Homma S, et al. Impaired flow-mediated vasodilatation is associated with increased left ventricular mass in a multiethnic population. The Northern Manhattan Study. Am J Hypertens 2010; 23: 413-9. https://doi.org/10.1038/ajh.2009.261 PMid:20057361 PMCid:PMC2847833 |
||||
| 12. Payne JR, James LE, Eleftheriou KI, Hawe E, Mann J, Stronge A, et al. The association of left ventricular mass with blood pressure, cigarette smoking and alcohol consumption; data from the LARGE Heart study. Int J Cardiol 2007; 120: 52-8. https://doi.org/10.1016/j.ijcard.2006.08.043 PMid:17079035 |
||||
| 13. Devereux RB, Roman MJ, Paranicas M, Lee ET, Welty TK, Fabsitz RR, et al. A population-based assessment of left ventricular systolic dysfunction in middle-aged and older adults: the Strong Heart Study. Am Heart J 2001; 141: 439-46. https://doi.org/10.1067/mhj.2001.113223 PMid:11231443 |
||||
| 14. Rosen BD, Saad MF, Shea S, Nasir K, Edvardsen T, Burke G, et al. Hypertension and smoking are associated with reduced regional left ventricular function in asymptomatic individuals: the Multi-Ethnic Study of Atherosclerosis. J Am Coll Cardiol 2006; 47: 1150-8. https://doi.org/10.1016/j.jacc.2005.08.078 PMid:16545644 |
||||
| 15. Dalen H, Thorstensen A, Romundstad PR, Aase SA, Stoylen A, Vatten LJ . Cardiovascular risk factors and systolic and diastolic cardiac function: a tissue Doppler and speckle tracking echocardiographic study. J Am Soc Echocardiogr 2011; 24: 322-32. https://doi.org/10.1016/j.echo.2010.12.010 PMid:21247733 |
||||
| 16. Kamimura D, Cain LR, Mentz RJ, White WB, Blaha MJ, DeFilippis AP, et al. Cigarette smoking and incident heart failure: insights from the Jackson Heart Study. Circ 2018; 137: 2572-82. https://doi.org/10.1161/CIRCULATIONAHA.117.031912 PMid:29661945 PMCid:PMC6085757 |
||||
| 17. Levy D, Garrison RJ, Savage DD, Kannel WB, Castelli WP. Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med 1990; 322: 1561-6. https://doi.org/10.1056/NEJM199005313222203 PMid:2139921 |
||||
| 18. Haider AW, Larson MG, Benjamin EJ, Levy D. Increased left ventricular mass and hypertrophy are associated with increased risk for sudden death. J Am Coll Cardiol 1998; 32: 1454-9. https://doi.org/10.1016/S0735-1097(98)00407-0 PMid:9809962 |
||||
| 19. Armstrong AC, Gidding S, Gjesdal O, Wu C, Bluemke DA, Lima JA. LV mass assessed by echocardiography and CMR, cardiovascular outcomes, and medical practice. J Am Coll Cardiol Img 2012; 5: 837-48. https://doi.org/10.1016/j.jcmg.2012.06.003 PMid:22897998 PMCid:PMC3501209 |
||||
| 20. Gidding SS. Controversies in the assessment of left ventricular mass. Hypertens 2010; 56: 26-8. https://doi.org/10.1161/HYPERTENSIONAHA.110.153346 PMid:20457999 |
||||
| 21. Ahmad A, Nawaz HM, Nevins L, Nawaz H. Longitudinal change in left ventricular hypertrophy in the elderly on antihypertensive therapy. Conn Med 2003; 67: 67-73. | ||||
| 22. Cicconetti P, Morelli S, Ottaviani L, Chiarotti F, De Serra C, De Marzio P, et al. Blunted nocturnal fall in blood pressure and left ventricular mass in elderly individuals with recently diagnosed isolated systolic hypertension. Am J Hypertens 2003; 16: 900-5. https://doi.org/10.1016/S0895-7061(03)01012-4 PMid:14573326 |
||||
| 23. Pontiroli AE, Pizzocri P, Saibene A, Girola A, Koprivec D, Fragasso G. Left ventricular hypertrophy and QT interval in obesity and in hypertension: effects of weight loss and of normalisation of blood pressure. Int J Obes 2004; 28: 1118-23. https://doi.org/10.1038/sj.ijo.0802733 PMid:15263923 |
||||
| 24. Davis CL, Kapuku G, Treiber FA, Kumar M, Snieder H. Insulin resistance syndrome and left ventricular mass in healthy young people. Am J Med Sci 2002; 324: 72-5. https://doi.org/10.1097/00000441-200208000-00005 PMid:12186110 |
||||
| 25. Marcus JT, DeWaal LK, Götte MJ, Van der Geest RJ, Heethaar RM, Rossum AV. MRI-derived left ventricular function parameters and mass in healthy young adults: relation with gender and body size. Int J Card Imaging 1999; 15: 411-9. https://doi.org/10.1023/A:1006268405585 PMid:10595407 |
||||
| 26. Bottini PB, Carr AA, Prisant LM, Flickinger FW, Allison JD, Gottdiener JS. Magnetic resonance imaging compared to echocardiography to assess left ventricular mass in the hypertensive patient. Am J Hypertens 1995; 8: 221-8. https://doi.org/10.1016/0895-7061(94)00178-E PMid:7794570 |
||||
| 27. Wu VC, Takeuchi M. Three-dimensional echocardiography: current status and real-life applications. Acta Cardiol Sin 2017; 33: 107-18. | ||||
| 28. Pouleur AC, de Waroux JL, Pasquet A, Gerber BL, Gerard O, Allain P, et al. Assessment of left ventricular mass and volumes by three-dimensional echocardiography in patients with or without wall motion abnormalities: comparison against cine magnetic resonance imaging. Heart 2008; 94: 1050-7. https://doi.org/10.1136/hrt.2007.123711 PMid:17974699 PMCid:PMC2564843 |
||||
| 29. Du Bois D, Du Bois EF. Clinical calorimetry: tenth paper a formula to estimate the approximate surface area if height and weight be known. Arch Intern Med 1916; 17: 863-71. https://doi.org/10.1001/archinte.1916.00080130010002 |
||||
| 30. Borland R, Yong HH, O'connor RJ, Hyland A, Thompson ME. The reliability and predictive validity of the Heaviness of Smoking Index and its two components: findings from the International Tobacco Control Four Country study. Nicotine Tob. Res 2010; 12 (Suppl 1) S45-50. https://doi.org/10.1093/ntr/ntq038 PMid:20889480 PMCid:PMC3307335 |
||||
| 31. Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2015; 16: 233-71. https://doi.org/10.1093/ehjci/jev014 PMid:25712077 |
||||
| 32. Devereux RB, Alonso DR, Lutas EM, Gottlieb GJ, Campo E, Sachs I, et al. Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol 1986; 57: 450-8. https://doi.org/10.1016/0002-9149(86)90771-X PMid:2936235 |
||||
| 33. Tei C, Ling LH, Hodge DO, Bailey KR, Oh JK, Rodeheffer RJ, et al. New Index of Combined Systolic and Diastolic Myocardial Performance: A Simple and Reproducible Measure of Cardiac Function-A Study in Normals and Dilated Cardiomyopathy. J Cardiol 1995; 26: 357-66. https://doi.org/10.1016/S0894-7317(05)80111-7 |
||||
| 34. Gerard O, Billon AC, Rouet JM, Jacob M, Fradkin M, Allouche C. Efficient model-based quantification of left ventricular function in 3-D echocardiography. IEEE Trans Med Imaging 2002; 21: 1059-68. https://doi.org/10.1109/TMI.2002.804435 PMid:12564874 |
||||
| 35. Dorosz JL, Lezotte DC, Weitzenkamp DA, Allen LA, Salcedo EE. Performance of 3-dimensional echocardiography in measuring left ventricular volumes and ejection fraction: a systematic review and meta-analysis. J Am Coll Cardiol 2012; 59: 1799-808. https://doi.org/10.1016/j.jacc.2012.01.037 PMid:22575319 PMCid:PMC3773600 |
||||
| 36. Pouleur AC, de Waroux JL, Pasquet A, Gerber BL, Gerard O, Allain P, et al. Assessment of left ventricular mass and volumes by three-dimensional echocardiography in patients with or without wall motion abnormalities: comparison against cine magnetic resonance imaging. Heart 2008; 94: 1050-7. https://doi.org/10.1136/hrt.2007.123711 PMid:17974699 PMCid:PMC2564843 |
||||
| 37. Takeuchi M, Nishikage T, Mor-Avi V, Sugeng L, Weinert L, Nakai H, et al. Measurement of left ventricular mass by real-time three-dimensional echocardiography: validation against magnetic resonance and comparison with two-dimensional and m-mode measurements. J Am Soc Echocardiogr 2008; 21: 1001-5. https://doi.org/10.1016/j.echo.2008.07.008 PMid:18765176 |
||||
| 38. Shah BN. Normal reference range values in adult echocardiography: Further evidence that race matters. Indian Heart J 2016; 68: 758-9. https://doi.org/10.1016/j.ihj.2016.04.004 PMid:27931541 PMCid:PMC5143823 |
||||
| 39. Løkke A, Lange P, Scharling H, Fabricius P, Vestbo J. Developing COPD: a 25 year follow up study of the general population. Thorax 2006; 61: 935-9. https://doi.org/10.1136/thx.2006.062802 PMid:17071833 PMCid:PMC2121175 |
||||
| 40. Simonneau G, Gatzoulis MA, Adatia I, Celermajer D, Denton C, Ghofrani A, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 2013; 62(25 Suppl): D34-D41. https://doi.org/10.1016/j.jacc.2013.10.029 PMid:24355639 |
||||
| 41. Freixa X, Portillo K, Paré C, Garcia-Aymerich J, Gomez FP, Benet M, et al. Echocardiographic abnormalities in patients with COPD at their first hospital admission. Eur Respir J 2013; 41: 784-91. https://doi.org/10.1183/09031936.00222511 PMid:23018914 |
||||
| 42. Baum C, Ojeda FM, Wild PS, Rzayeva N, Zeller T, Sinning CR, et al. Subclinical impairment of lung function is related to mild cardiac dysfunction and manifest heart failure in the general population. Int J Cardiol 2016; 218: 298-304. https://doi.org/10.1016/j.ijcard.2016.05.034 PMid:27240155 |
||||
| 43. Manisty CH, Francis DP. Ejection fraction: a measure of desperation?. Heart 2008; 94: 400-1. https://doi.org/10.1136/hrt.2007.118976 PMid:18347366 |
||||
| 44. Yeon SB, Salton CJ, Gona P, Chuang ML, Blease SJ, Han Y, et al. Impact of age, sex, and indexation method on MR left ventricular reference values in the Framingham Heart Study offspring cohort. J Magn Reson Imaging 2015; 41: 1038-45. https://doi.org/10.1002/jmri.24649 PMid:24817313 PMCid:PMC4248013 |
||||
| 45. Kalam K, Otahal P, Marwick TH. Prognostic implications of global LV dysfunction: a systematic review and meta-analysis of global longitudinal strain and ejection fraction. Heart 2014; 100: 1673-80. https://doi.org/10.1136/heartjnl-2014-305538 PMid:24860005 |
||||
| 46. Omvik P. How smoking affects blood pressure. Blood Press 1996; 5: 71-7. https://doi.org/10.3109/08037059609062111 PMid:9162447 |
||||
| 47. Mahmud A, Feely J. Effect of smoking on arterial stiffness and pulse pressure amplification. Hypertension 2003; 41: 183-7. https://doi.org/10.1161/01.HYP.0000047464.66901.60 PMid:12511550 |
||||
| 48. Yap SC, van Geuns RJ, Nemes A, Meijboom FJ, McGhie JS, Geleijnse ML, et al. Rapid and accurate measurement of LV mass by biplane real-time 3D echocardiography in patients with concentric LV hypertrophy: comparison to CMR. Eur J Echocardiogr 2008; 9: 255-60. https://doi.org/10.1016/j.euje.2007.03.037 PMid:17604225 |
||||
| 49. Shimada YJ, Shiota T. Meta-analysis of accuracy of left ventricular mass measurement by three-dimensional echocardiography. Am J Cardiol 2012; 110: 445-52. https://doi.org/10.1016/j.amjcard.2012.03.046 PMid:22541420 |
||||
| 50. Shimada YJ, Shiota T. A meta-analysis and investigation for the source of bias of left ventricular volumes and function by three-dimensional echocardiography in comparison with magnetic resonance imaging. Am J Cardiol 2011; 107): 126-38. https://doi.org/10.1016/j.amjcard.2010.08.058 PMid:21146700 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

