Patient comfort and access site outcomes with a screw-based mechanical radial compression device following transradial coronary intervention: A quasi-randomized study
ORIGINAL RESEARCH ARTICLE
Patient comfort and access site outcomes with a screw-based mechanical radial compression device following transradial coronary intervention: A quasi-randomized study
Article Summary
- DOI: 10.24969/hvt.2026.637
- CARDIOVASCULAR DISEASES
- Published: 15/03/2026
- Received: 11/02/2026
- Revised: 13/03/2026
- Accepted: 14/03/2026
- Views: 513
- Downloads: 270
- Keywords: Radial artery, percutaneous coronary intervention, hemostasis, a screw-based device
Address for Correspondence: Khuong Dang Tran, Apartment C06.07, The Privia Khang Dien Residence, 321 An Duong Vuong Street, An Lac Ward, Binh Tan District, Ho Chi Minh City, Vietnam
Email: khuong.td2@umc.edu.vn Mobile: +84 0352731868
ORCID: Hoa Tran: 0000-0001-8393-6442; Dat Minh Nguyen: 0009-0009-1207-1379; Nhut Quang Le: 0009-0008-0971-2190; Khoa Ba Tran: 0009-0007-8658-6813; Hung Quoc Pham: 0009-0007-8453-3670; Tan Minh Nguyen: 0009-0003-1458-8917; Khuong Dang Tran: 0009-0003-7709-9088
Hoa Tran1a,2, Dat Minh Nguyen1a, Nhut Quang Le1a, Khoa Ba Tran1a, Hung Quoc Pham1a, Tan Minh Nguyen1a, Khuong Dang Tran1b
1aEndovascular Intervention Unit, 1bCardiology Department, University Medical Center Ho Chi Minh City, Ho Chi Minh City, Vietnam
2Department of Internal Medicine, University of Medicine and Pharmacy at Ho Chi Minh City, Ho Chi Minh City, Vietnam
Abstract
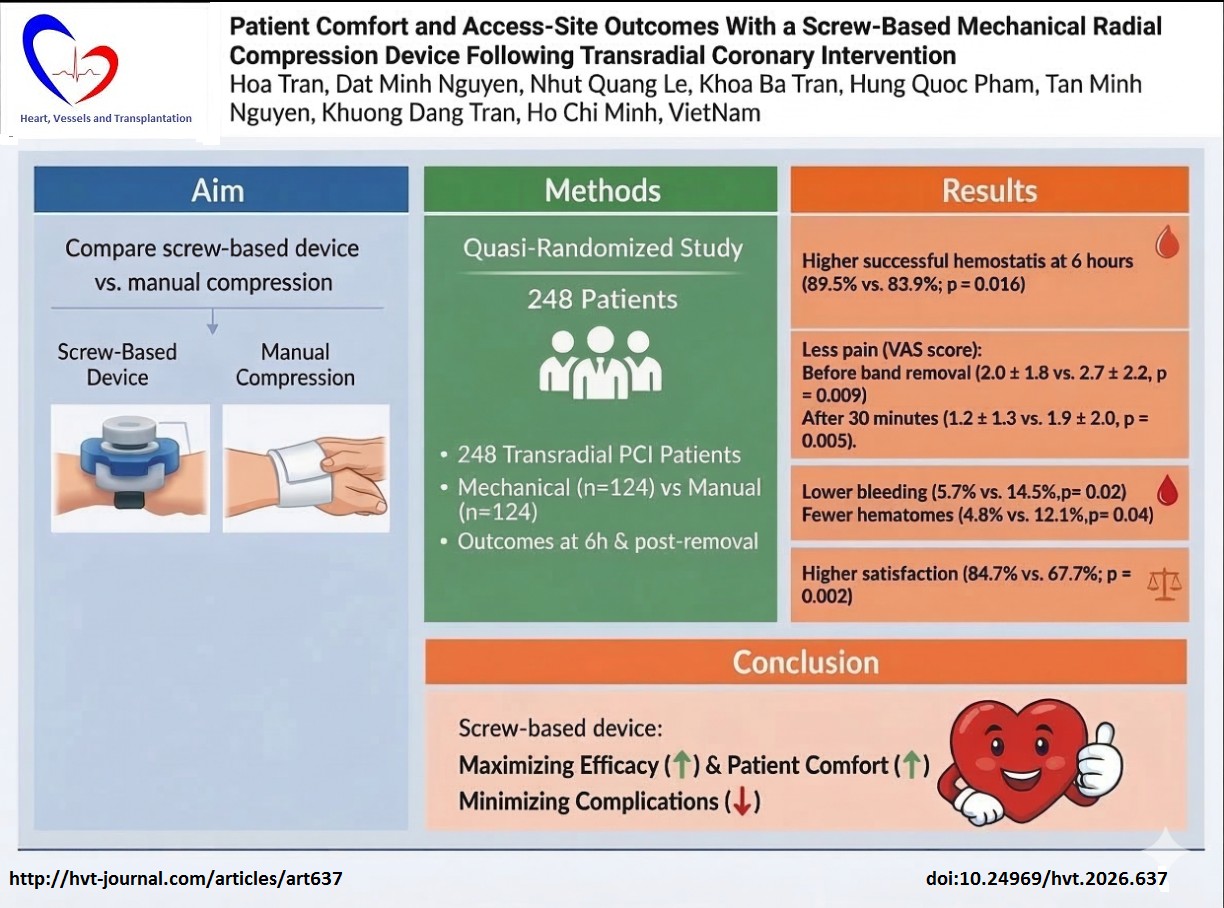
Objective: To evaluate whether a screw-based mechanical radial compression device improves hemostasis success, patient-reported pain and satisfaction compared with conventional manual compression after transradial coronary intervention.
Methods: A prospective, quasi-randomized trial was conducted in 248 patients undergoing percutaneous coronary intervention (PCI). Participants were allocated to either mechanical (n = 124) or manual groups (n = 124) in an 1:1 ratio. The primary outcome was successful hemostasis at 6 hours after compression. Bleeding and hematoma were assessed within 24 hours after the procedure. Pain intensity, discomfort, and overall satisfaction, were also recorded. Between-group comparisons were summarized using risk ratios with 95% confidence intervals.
Results: Successful hemostasis at 6 hours was significantly higher in the mechanical group compared with the manual group (89.5% vs. 83.9%; p = 0.016). The mechanical group had lower access-site bleeding (5.7% vs. 14.5%, p = 0.02) and fewer hematomas (4.8% vs. 12.1%, p = 0.04). Pain scores were significantly lower with the screw-based device both before (2.0 (1.8) vs. 2.7 (2.2), p = 0.009) and 30 minutes after band removal (1.2 (1.3) vs. 1.9 (2.0), p = 0.005). Patients also reported less discomfort (25.0% vs. 37.1%; p = 0.040) and higher satisfaction (84.7% vs. 67.7%; p = 0.002) in mechanical group. The were no complications related to a screw-based device use.
Conclusions: Compared with manual compression, a screw-based device provides superior hemostatic efficacy and reduces access-site complications while significantly improving patient experience, with lower pain during compression and after band removal.
Key words: Radial artery, percutaneous coronary intervention, hemostasis, a screw-based device
![]()

Introduction
Cardiovascular disease remains the leading cause of death worldwide. In 2022, an estimated 19.8 million people died from cardiovascular diseases, and ~85% of these deaths were attributable to heart attack and stroke (1). Against this background, percutaneous coronary intervention (PCI) remains a cornerstone therapy for coronary artery disease and acute coronary syndromes (2). Over the past decade, transradial access (TRA) has become the preferred approach for coronary angiography and PCI in many centers because it reduces access-site bleeding and vascular complications compared with femoral access and in acute coronary syndrome populations, it has been associated with improved clinical outcomes (2-4). However, optimal post-procedural hemostasis after radial sheath removal remains clinically important. Access-site bleeding, hematoma, and radial artery occlusion (RAO) are among the most relevant local complications; RAO is frequently clinically silent but may preclude future use of the radial artery for repeat coronary procedures (5).
RAO remains a relevant access-site complication after transradial coronary procedures. In the multicenter randomized MEMORY trial comparing manual versus mechanical radial compression after transradial coronary angiography, early RAO occurred at similar rates (12% with manual vs. 8% with mechanical compression; p = 0.176), suggesting broadly comparable RAO risk with a small numerical trend favoring mechanical compression(6). Consistently, a prospective randomized study comparing a compression band device with manual gauze compression also found no significant difference in early RAO evaluated by duplex ultrasonography within 24–72 hours (1.47% vs. 2.9%; P = 1.000)(7). Collectively, these data indicate that when contemporary strategies to preserve radial flow are applied, manual and mechanical approaches generally yield comparable RAO outcomes, with potential incremental benefit from protocols that facilitate consistent, titratable compression.
However, evidence on patient experience with adjustable screw/rotary-type radial compression bands remains limited and methodologically heterogeneous. In the largest randomized comparison that included a rotary compression pad device (n = 1,650), Cong et al. (8) reported a statistically significant difference in ‘discomfort level’ across hemostasis strategies, with the rotary compression device demonstrating a lower overall discomfort rate compared with conventional elastic bandage compression. However, the comfort assessment was essentially a single, global tolerance metric rather than a clearly defined, multidimensional patient-reported outcome (8). More granular comfort data in screw/rotatory-device settings largely come from small, open-label trials. Kang et al. (9) studied 95 patients receiving rotatory compression devices with or without a hemostasis pad and used a simple numeric discomfort rating; importantly, the trial was stopped prematurely before planned enrollment, and safety endpoints (including vascular complications) were too infrequent to detect meaningful between-group differences. Likewise, a prospective randomized study comparing a novel device (pressure-controlled, focused-compression mechanical radial hemostasis device) with the pneumatic TR Band enrolled only 60 patients; although pain and swelling favored the novel device, the small sample size and device-specific design limit generalizability (10).
Moreover, manual gauze compression is still widely used in many developing countries because of its low cost. Therefore, comparison against manual compression remains highly relevant to real-world practice in resource-constrained settings. Second, pneumatic compression systems are not always the most feasible option in high-volume centers with limited resources. Randomized data comparing the screw-based VasoStat device with the pneumatic TR Band showed that the screw-based device required fewer device manipulations while maintaining effective hemostasis (11). Therefore, in settings with limited financial and human resources, a screw-based device may represent a pragmatic intermediate option: more standardized and controllable than manual gauze compression, yet potentially less resource-intensive and more affordable than pneumatic systems. At the same time, direct comparisons between screw/rotary devices and conventional manual gauze compression remain limited in the literature. For this reason, we believe that study comparing screw/rotary devices and manual compression adds useful evidence for centers seeking a practical alternative to manual gauze compression without increasing workflow complexity
Given that major RAO-related questions have been extensively explored in prior work, in contrast, the evidence base for patient-centered outcomes remains comparatively weak, especially for adjustable screw/rotary-type compression systems, where comfort may plausibly differ despite similar vascular safety.
Therefore, we conducted a study to compare an adjustable screw-based mechanical compression band with conventional manual compression after transradial coronary intervention, prioritizing patient experience (pain, discomfort, and satisfaction) while also documenting hemostatic success and access-site complications.
Methods
Study design and population
This prospective, controlled, quasi-randomized clinical trial was conducted at the Endovascular Intervention Unit, in University Medical Center Ho Chi Minh City, between August 2025 and December 2025. The study enrolled consecutive patients undergoing transradial coronary intervention (coronary angiography and PCI) via the radial artery. Eligible participants were adults (≥18 years) undergoing a transradial coronary procedure. Exclusion criteria included failed TRA; skin lesions, scars, or a history of wrist fracture at the access site; previously diagnosed arteriovenous fistula in the arm; technical difficulties preventing completion of the procedure according to protocol; and inability to communicate and/or to cooperate adequately during the procedure.
Participants were allocated in an approximate 1:1 ratio using a quasi-randomized, day-of-week allocation scheme. Specifically, patients undergoing transradial PCI on Mondays, Wednesdays, and Fridays were assigned to the screw-based mechanical radial compression device group (mechanical group), whereas those treated on Tuesdays, Thursdays, and Saturdays were assigned to the conventional manual compression group (manual group). As allocation was determined by the day of the procedure, formal allocation concealment was not feasible, and blinding of treating staff was not possible due to the nature of the interventions. To minimize expectation-related bias, participants were not explicitly informed which method corresponded to “manual” versus “screw-based” compression during outcome assessment
The study was reviewed and approved by the Institutional Review Board/Ethics Committee of the University Medical Center Ho Chi Minh City (approval No. 38/GCN-HĐĐĐ). The study was conducted in accordance with the ethical principles of the Declaration of Helsinki as revised in 2024.
Participation was voluntary, and written informed consent was obtained after participants received full information regarding study objectives, procedures, and potential risks. Participant confidentiality was strictly protected, and data were used solely for research purposes.
Baseline variables
Baseline variables included demographic characteristics (age, sex, and body mass index, BMI), cardiovascular risk factors (hypertension, diabetes mellitus, dyslipidemia, and current smoking status), prior PCI, and procedural characteristics such as sheath size, procedure time, and procedural heparin dose.
Intervention and compression device
All procedures were performed using standard transradial techniques. Radial sheath size and procedural anticoagulation were determined by the operator according to institutional practice (e.g., sheath size, unfractionated heparin dose; use of glycoprotein IIb/IIIa inhibitors). After completion of the coronary procedure, the arterial sheath was removed and hemostasis was achieved using one of two strategies.
In the mechanical group, the device (Mostar™, USM Healthcare, Vietnam) was positioned directly over the puncture site at the time of sheath removal and secured around the wrist. According to the manufacturer, the device is intended for right or left radial artery hemostasis only and is not designed for distal radial or ulnar access. Compression was adjusted to achieve local hemostasis while maintaining ulnar artery perfusion, adhering to a patent hemostasis principle. The initial compression level was maintained for approximately 2-3 hours, then reduced according to the standardized nursing care protocol. The device was removed at 6 hours provided there was no bleeding or access-site complication (Fig. 1).
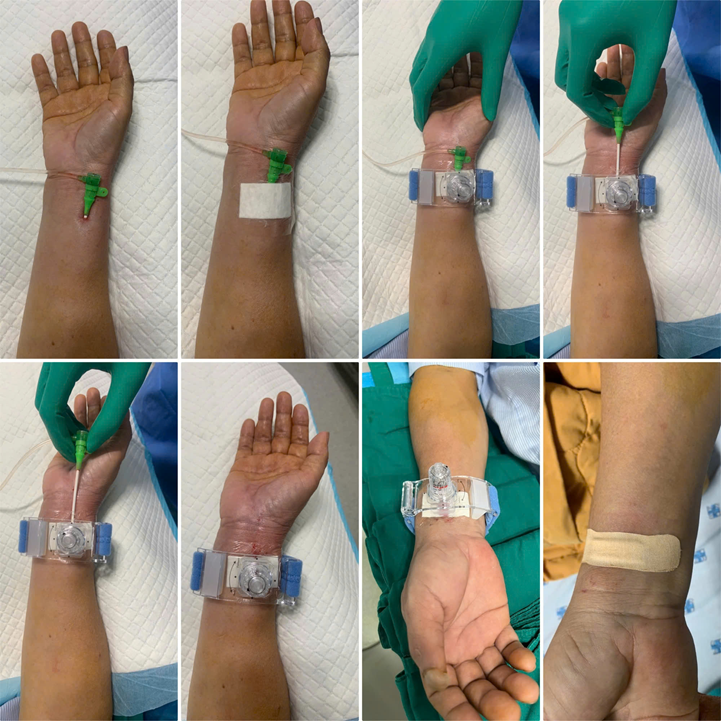
Figure 1. Application and stepwise deflation of an adjustable screw-based mechanical radial compression device after transradial coronary intervention
In the manual compression group (gauze), a sterile 2 × 2 cm gauze pad was placed at the puncture site, followed by localized compression using elastic adhesive tape applied according to standard technique. The initial degree of compression was adjusted to achieve hemostasis while maintaining adequate hand perfusion according to the principle of patent hemostasis. In both groups, hand circulation was assessed using pulse oximetry plethysmography during compression. In addition, a modified Allen test was routinely performed before the procedure to confirm adequate collateral circulation of the hand. Throughout compression, ulnar artery perfusion was ensured to minimize ischemic risk. The compression dressing was removed at 6 hours if no bleeding, hematoma, or other access-site complications were observed (Fig. 2).
Figure 2. Conventional manual compression using gauze and elastic adhesive bandage after transradial coronary intervention
No crossover between the two treatment groups occurred in this study. All patients received the hemostasis method according to their initially assigned group throughout the observation period. If complete hemostasis was not achieved at the planned 6-hour compression removal, compression was continued using the same technique previously assigned. Additional compression was applied for 30–60 minutes as needed until complete hemostasis was achieved.
Patients were routinely monitored with bedside monitoring, including oxygen saturation, during the first 6 hours after the procedure. This allowed continuous assessment of hand perfusion during radial compression. In addition, radial pulse and clinical signs of hand ischemia, such as pain or numbness, were evaluated during follow-up. No adjunctive hemostatic patches were used in this study.
Outcome variables
Pain outcomes were assessed using a visual analog scale (VAS, 1–10) supported by a facial-expression chart (faces graded from 1 to 10) to facilitate patient self-reporting. VAS pain scores were recorded immediately before device/dressing removal and 30 minutes after removal. All pain assessments were performed by the same technician. Access-site complications included bleeding and hematoma. Minor bleeding was defined as any visible bleeding or oozing during compression and/or at the time of dressing/device removal. Hematoma was assessed at 24 hours after the procedure and graded according to the EASY hematoma classification (grades I–V), reflecting hematoma extent and severity.
Statistical analysis
Data were entered, managed, and analyzed using Stata version 17. Univariate analyses were performed to describe and compare demographic, clinical characteristics, and study outcomes between groups. For normally distributed continuous variables, between-group differences were assessed using the independent t-test; for non-normally distributed continuous variables, the Mann–Whitney U test was used. Categorical variables were compared using the Chi-squared test or Fisher’s exact test as appropriate.
For binary outcomes, intervention effects were summarized as risk ratios (RRs) with 95% confidence intervals (95% CIs). A two-sided p value < 0.05 was considered statistically significant.
Results
A total of 248 patients were included in the final analysis, of whom 124 were assigned to the mechanical group and 124 to the manual group, ensuring balanced allocation and comparability between the two treatment arms (Fig. 3).
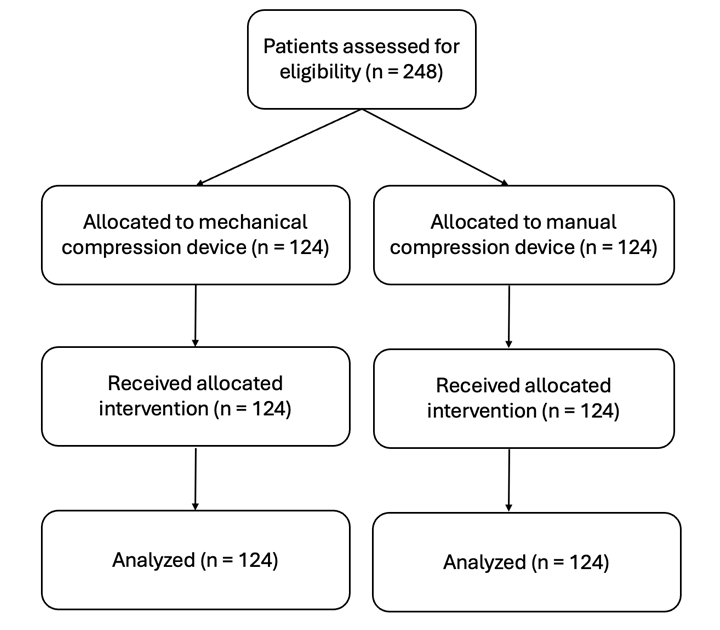
Figure 3. Flowchart of patients` enrollment to the study
Clinical characteristics
The mean age of the overall study population was 63.0(8.9) years, and 174 patients (70.2%) were male. Comparison results of baseline demographic characteristics, cardiovascular risk factors, and procedural variables are presented in Table 1.
No statistically significant differences were observed between the mechanical and manual groups in baseline characteristics or clinical risk factors, including age, sex, BMI, prevalence of hypertension, diabetes mellitus, dyslipidemia, current smoking status, and history of PCI.
Likewise, procedural parameters such as sheath size distribution (6F vs. 7F), procedure time, and heparin dose did not differ significantly between groups. All comparisons yielded p values > 0.05, confirming a high degree of baseline homogeneity.
|
Table 1. Baseline characteristics of the study population |
|||||
|
Variables |
Total |
Mechanical group (n = 124) |
Manual group (n = 124) |
p |
|
|
Age, years |
63.0 (8.9) |
63.3 (8.8) |
62.6 (9.1) |
0.52 |
|
|
Male n (%) |
174 (70.2) |
88 (71.0) |
86 (69.4) |
0.78 |
|
|
BMI, kg/m², |
23.5 ± 3.0 |
23.6 ± 2.8 |
23.4 ± 3.2 |
0.58 |
|
|
Risk factors for CAD, n (%) |
|||||
|
Hypertension Diabetes mellitus Dyslipidemia Smoking, n (%) |
161 (64.9) 95 (38.3) 119 (48.0) 65 (26.2) |
79 (63.7) 45 (36.3) 60 (48.4) 36 (29.0) |
82 (66.1) 50 (40.3) 66 (53.23) 29 (23.4) |
0.68 0.51 0.44 0.31 |
|
|
Previous PCI, n (%) |
25 (10.1) |
9 (7.3) |
16 (12.9) |
0.14 |
|
|
Sheath size, n (%) |
|||||
|
6F 7F |
149 (60.1) 99 (39.9) |
73 (58.8) 51 (41.2) |
76 (61.3) 48 (38.7) |
0.69 |
|
|
Procedure time, min |
55.4 (7.4) |
54.6 (7.3) |
56.0 (7.4) |
0.14 |
|
|
Heparin, IU |
4408.3 (657.5) |
4408.8 (627.0) |
4363.7 (688.5) |
0.59 |
|
|
Data are presented as mean (standard deviation) and number (%) Chi-square or Fischer exact test, t-test for independent samples or Mann Whitney U BMI – body mass index, CAD – coronary artery disease, PCI – percutaneous coronary intervention |
|||||
Efficacy of mechanical vs manual compression after PCI
Hemostatic efficacy of the two compression methods is summarized in Table 2. Overall hemostasis success was high in both groups. Notably, the rate of successful hemostasis at 6 hours was significantly higher in the mechanical group than in the manual group (89.5% vs. 83.9%; p = 0.016).
Access-site complications occurred at a relatively low frequency in both groups; however, the incidence was significantly lower in the Mostar group (Table 2). Specifically, the overall rate of access-site bleeding was also significantly lower in the mechanical group (5.7% vs. 14.5%; RR = 0.39; 95% CI: 0.16–0.89; p = 0.02). Most bleeding events were classified as minor oozing without clinically significant consequences; no major bleeding was recorded in both groups. All hematomas were graded as EASY grade I, and the incidence of hematoma was significantly lower in the Mostar group (RR = 0.40; 95% CI: 0.16–0.99; p = 0.04).
|
Table 2. Clinical outcomes and patient-reported measures |
||||
|
Variables |
Mechanical group (n = 124) |
Manual group n = 124) |
RR (95% CI) |
p |
|
Successful hemostasis, n (%) |
111 (89.5) |
97 (83.9) |
1.14 (1.02 – 1.28) |
0.016 |
|
Access-site complications, n (%) |
9 (7.3) |
22 (17.7) |
0.41 (0.19–0.85) |
0.013 |
|
Minor bleeding, n (%) |
7 (5.7) |
18 (14.5) |
0.39 (0.16–0.89) |
0.020 |
|
Hematoma, n(%) |
6 (4.8) |
15 (12.1) |
0.4 (0.16–0.99) |
0.040 |
|
Hematoma classification |
||||
|
Grade 1 Grade 2 Grade 3 Grade 4 Grade 5 |
6 (100) 0 0 0 0 |
15 (100) 0 0 0 0 |
0.040 |
|
|
VAS before band removal, points |
2.0 ± 1.8 |
2.7 ± 2.2 |
0.009 |
|
|
VAS 30 minutes after removal, points |
1.2 ± 1.3 |
1.9 ± 2.0 |
0.005 |
|
|
Discomfort, n(%) |
31 (25) |
46 (37.1) |
0.67 (0.45–0.98) |
0.040 |
|
Satisfaction, n(%) |
105 (84.7) |
84 (67.7) |
1.25 (1.08–1.44) |
0.002 |
|
Data are presented as mean (standard deviation) and number (%) Chi-square or Fischer exact test, t-test for independent samples or Mann Whitney U test CI – confidence interval, RR – relative risk, VAS – visual analog score |
||||
Patient-reported pain, discomfort, and satisfaction are presented in Table 2. Before device or band removal, the mean VAS pain score in the mechanical group was lower than VAS pain score in the manual group. At 30 minutes after removal, pain levels decreased in both groups but remained significantly lower in the mechanical group. Differences in pain scores at both time points were statistically significant (p<0.05). In addition, the proportion of patients reporting discomfort during compression was significantly lower in the mechanical group (p=0.04). Patient satisfaction was also significantly higher with the mechanical group (p=0.002).
Discussion
In this prospective, quasi-randomized (systematically allocated) study comparing screw-based mechanical radial compression device with manual compression after PCI, the mechanical group demonstrated superior hemostatic success and patient comfort. Specifically, hemostasis at 6 hours was achieved in 89.5% of patients with the screw device versus 83.9% with manual gauze (p=0.016). Correspondingly, acute access-site bleeding (5.7% vs 14.5%) and EASY grade I hematoma (4.8% vs 12.1%) were significantly less frequent in the mechanical group. These improvements are accompanied by marked patient-centered benefits: patients with the screw device reported lower pain scores both immediately before and 30 minutes after band removal, and less overall discomfort (25.0% vs 37.1%; p=0.04). They also expressed higher satisfaction with the hemostasis process. In aggregate, the mechanical compression yielded more efficient hemostasis, fewer complications and more comfort – a finding that reflects both the device’s controllability and the advantages of patent hemostasis.
Mechanistically, the pain reduction likely stems from the focused, adjustable pressure of the screw-based band. The device applies compression directly over the puncture site with a calibrated force, avoiding the broad circumferential pressure of a gauze-and-tape wrap. Diffuse manual compression can inadvertently compress surrounding nerves and vessels, leading to ischemic discomfort or neuropathic pain. By contrast, a screw mechanism permits gradual, localized compression while preserving flow through the ulnar artery, minimizing collateral tissue trauma. This targeted approach would be expected to reduce both acute pain during device application and sustained pain intensity after device removal. Our data support this: mean visual analog pain scores were lower in the mechanical group before removal (2.0 vs 2.7) and remained lower at 30 minutes post-removal (1.2 vs 1.9; both p<0.01). Post-removal pain is a clinically relevant and often underappreciated component of transradial care because it can impair comfort, delay mobilization, and negatively influence overall treatment experience. In a large cohort of 1,706 patients, post-procedural arm pain (assessed the day after a transradial procedure) occurred in 4.5%, and prolonged hemostasis compression >4 hours was the strongest predictor (OR 29.47; p<0.001), highlighting that not only the device type but also the intensity and duration of compression substantially influence pain outcomes (12). Importantly, protocol optimization appears to meaningfully reduce pain: in a controlled trial evaluating early versus typical TR Band deflation, early deflation was associated with less pain (p=0.003) without a significant increase in hematoma or RAO, supporting the concept that minimizing sustained external pressure can improve patient comfort (13). This provides a mechanistic framework to interpret our findings—our screw-based device, by enabling fine, incremental pressure adjustment, likely contributed to the lower pain scores observed after band removal in our cohort. Beyond timing, device design also matters among mechanical strategies: compared with pneumatic TR Band compression, a randomized trial found that VasoStat (a screw-based mechanical device) resulted in significantly lower patient discomfort and fewer device manipulations, suggesting that localized, controllable compression may be better tolerated than broader pneumatic bladder pressure (11).
Crucially, the mechanical device also enhanced efficiency of hemostasis. In our research, a higher proportion of patients in the screw-device arm were fully hemostatic at the 6-hour mark, implying more patients could be released on schedule and fewer required extended compression or hospitalization. In practical terms, this reduces nursing workload and may shorten hospital stay.
This is consistent with prior studies showing dedicated radial compression systems shorten time-to-hemostasis. For example, Cong et al. (8) found that both pneumatic and rotary compression devices achieved hemostasis significantly faster than a simple pressure dressing (average ~4–5 h vs ~5–6 h; p<0.0001).
In our trial, the screw device’s adjustable pressure likely helped attain rapid hemostasis: unlike gauze, the band can be tensioned just enough to stop bleeding and then loosened as swelling resolves. By avoiding overly tight or prolonged compression, it both secured the puncture and prevented the rebound oozing seen when wraps are removed.
We note one caution: in the MEMORY trial the mechanical (TR Band) group actually had a much longer compression time than manual (119 vs 22 min), but that protocol did not target patent hemostasis in the same way and is not directly comparable (6).
Our protocol, which maintained flow while compressing, appears effective: the higher 6-hour hemostasis rate with the screw device suggests that in real-world use it can reliably stop bleeding without excessive delay.
The reduction in bleeding and bruising in the mechanical group is also notable. Consistent pressure applied by the screw head seems to stabilize the puncture more evenly, whereas manual methods may slip or require very tight bandaging. In our data, minor bleeding was more than twice as common with gauze (14.5% vs 5.7%, p=0.02) and EASY grade I hematomas likewise (12.1% vs 4.8%, p=0.04). Other investigators have observed similar trends: in Cong’s series, simple elastic dressings were associated with more puncture-site ooze (8.2%) than pneumatic or rotary devices (≈5.1%) (8). The screw device’s ability to hold a constant, localized pressure likely accounts for this: it avoids the “over-compression” needed to achieve hemostasis with gauze (which can cause hematoma once loosened) and reduces the need for reapplication that might tear delicate post-angioplasty tissues.
Some contemporary radial compression protocols allow earlier device removal, sometimes beginning with gradual deflation at approximately 60–90 minutes. However, it is important to note that many studies evaluating screw-based compression devices have primarily included patients undergoing diagnostic coronary angiography, where procedural anticoagulation is lower and smaller sheath sizes are typically used (13, 14). In contrast, the majority of patients in our study underwent transradial PCI and received dual antiplatelet therapy, with larger sheath sizes and higher levels of procedural anticoagulation. These factors are known to increase the risk of access-site bleeding and may justify a more conservative compression strategy. To ensure consistent post-procedural management in this setting, a standardized compression protocol with device removal at 6 hours was adopted rather than repeated device adjustments every 30 minutes to have shorter device removal time. We believe that this approach reflects real-world workflow in many high-volume centers, where similar challenges related to patient volume and resource allocation are encountered. Therefore, our study may provide practical evidence for institutions with comparable clinical settings when considering the use of screw-based compression devices.
Study limitations
Our study did not directly assess RAO. RAO was not systematically assessed using Doppler ultrasound, which is a limitation. However, prior evidence suggests that devices like the mechanical device do not increase RAO risk and may even improve it when patent hemostasis is used. So existing data indicate that safety is comparable (8, 11).
Other limitations merit mention. We deviated from an ideal multi-step deflation protocol due to patient volume: rather than gradually reducing pressure hourly, we performed a single pressure reduction at 3 hours. This simplification may have influenced pain or hemostasis, since slow staged deflation is recommended to maximize comfort and safety. Additionally, the quasi-randomized design and lack of operator blinding could introduce bias, although baseline characteristics were well balanced. In this study, satisfaction and discomfort were captured using a single binary (yes/no) question, rather than a validated, multidimensional patient-reported outcome instrument. This approach may have limited sensitivity to detect nuanced differences across domains such as functional restriction of the wrist/hand, sleep disturbance. Despite these caveats, the consistency of our findings across objective (bleeding) and subjective (pain/satisfaction) measures supports a true advantage of the screw-based device.
Conclusion
In conclusion, screw-based mechanical radial compression device outperformed standard manual gauze compression in terms of patient experience and access-site outcomes. By delivering titratable, localized pressure, the device achieved higher first-attempt hemostasis and significantly reduced patient pain and discomfort. These findings reinforce evidence that dedicated radial compression bands can improve efficiency and patient comfort after coronary procedures. Adoption of such mechanical devices may therefore enhance workflow and patient satisfaction. Our results suggest that screw-type radial compression is a promising strategy for improving post-PCI care.
Ethics: The study was reviewed and approved by the Institutional Review Board/Ethics Committee of the University Medical Center Ho Chi Minh City (approval No. 38/GCN-HĐĐĐ). The study was conducted in accordance with the ethical principles of the Declaration of Helsinki as revised in 2024. Participation was voluntary, and written informed consent was obtained after participants received full information regarding study objectives, procedures, and potential risks. Participant confidentiality was strictly protected, and data were used solely for research purposes.
Peer-review: External and internal
Conflict of interest: None to declare
Authorship: H.T. - the original conception of the study idea; K.D.T. was responsible for manuscript preparation and drafting; D.M.N. contributed to data collection and statistical analysis; N.Q.L. K.B.T., H.Q.P. and T.M.N. assisted in patient recruitment and data acquisition. All authors critically read and approved revised version, thus equally contributed to preparation of manuscript and fulfilled all authorship criteria
Acknowledgements: The authors would like to acknowledge the contributions of Hoa Tran for original conception of the study idea.
Funding: None to declare. No external funding was received for this study. The authors also declare that no commercial organization or company provided any financial support, equipment, or materials for the completion of this research or manuscript.
Statement on A.I.-assisted technologies use: Artificial intelligence tools were used only for language editing. All scientific content, data analysis, and interpretation were performed by the authors, who take full responsibility for the integrity and accuracy of the work.
Data and material availability: The de-identified individual participant data that support the findings of this study are available from the corresponding author upon reasonable request.
Any share should be in frame of fair use, with acknowledgement of source and/ or collaboration
References
| 1.Mensah George A, Fuster V, Roth Gregory A. A heart-healthy and stroke-free world. J Am Coll Cardiol 2023; 82: 2343-9. https://doi.org/10.1016/j.jacc.2023.11.003 PMid:38092508 |
||||
| 2.Byrne RA, Rossello X, Coughlan JJ, Barbato E, Berry C, Chieffo A, et al. 2023 ESC Guidelines for the management of acute coronary syndromes: Developed by the task force on the management of acute coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J 2023; 44: 3720-826. https://doi.org/10.1093/eurheartj/ehad191 PMid:37622654 |
||||
| 3.Lawton JS, Tamis-Holland JE, Bangalore S, Bates ER, Beckie TM, Bischoff JM, et al. 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization. J Am Coll Cardiol 2022; 79: e21-e129. https://doi.org/10.1016/j.jacc.2021.09.006 PMid:34895950 |
||||
| 4.Gargiulo G, Giacoppo D, Jolly SS, Cairns J, Le May M, Bernat I, et al. Effects on mortality and major bleeding of radial versus femoral artery access for coronary angiography or percutaneous coronary intervention: Meta-analysis of individual patient data from 7 multicenter randomized clinical trials. Circulation 2022; 146: 1329-43. https://doi.org/10.1161/CIRCULATIONAHA.122.061527 PMid:36036610 |
||||
| 5.Rashid M, Kwok CS, Pancholy S, Chugh S, Kedev SA, Bernat I, et al. Radial artery occlusion after transradial interventions: a systematic review and meta‐analysis. J Am Heart Assoc XXXX; 5: e002686. https://doi.org/10.1161/JAHA.115.002686 PMid:26811162 PMCid:PMC4859386 |
||||
| 6.Petroglou D, Didagelos M, Chalikias G, Tziakas D, Tsigkas G, Hahalis G, et al. Manual versus mechanical compression of the radial artery after transradial coronary angiography: The MEMORY multicenter randomized trial. JACC Cardiovasc Interv 2018; 11: 1050-8. https://doi.org/10.1016/j.jcin.2018.03.042 PMid:29880098 |
||||
| 7.Ramos HJR, Marcelo J. Comparison of radial artery occlusion occurrence (RAO) between compression band device and manually applied gauze compression after transradial coronary procedure, a PROBE study. Eur J Prev Cardiol 2023; 30(Suppl_1): zwad125.069. https://doi.org/10.1093/eurjpc/zwad125.069 |
||||
| 8.Cong X, Huang Z, Wu J, Wang J, Wen F, Fang L, et al. Randomized comparison of 3 hemostasis techniques after transradial coronary intervention. J Cardiovasc Nurs 2016; 31: 445-51. https://doi.org/10.1097/JCN.0000000000000268 PMid:26002786 |
||||
| 9.Kang S-H, Han D, Kim S, Yoon C-H, Park J-J, Suh J-W, et al. Hemostasis pad combined with compression device after transradial coronary procedures: A randomized controlled trial. PLOS ONE 2017; 12: e0181099. https://doi.org/10.1371/journal.pone.0181099 PMid:28742134 PMCid:PMC5524363 |
||||
| 10.Wu B, Zhang R, Liang C, Zhang C, Qin G. Study on the safety of the new radial artery hemostasis device. J Interv Cardiol 2022; 2022: 2345584. https://doi.org/10.1155/2022/2345584 PMid:35463209 PMCid:PMC9005317 |
||||
| 11.Minor RL Jr, Maley T, Jenkins D, Li YH. Randomized trial of vasostat versus tr band following radial artery access for catheterization procedures. J Invasive Cardiol 2021; 33: E84-e90. | ||||
| 12.Dharma S, Kedev S, Patel T, Gilchrist IC, Rao SV. The Predictors of post-procedural arm pain after transradial approach in 1706 patients underwent transradial catheterization. Cardiovasc Revasc Med 2019; 20: 674-7. https://doi.org/10.1016/j.carrev.2018.09.006 PMid:30245153 |
||||
| 13.Kheirabad MK, Mohebbi Z, Kalyani MN, Kojuri J. Vascular outcomes of early deflation of radial artery band following coronary angiography: A controlled clinical trial. J Vasc Nurs 2023; 41: 56-61. https://doi.org/10.1016/j.jvn.2023.04.001 PMid:37356871 |
||||
| 14.Riyami HA, Riyami AA, Nadar SK. Comparison between two protocols for deflation of the TR band following coronary procedures via the radial route. J Saudi Heart Assoc 2020; 32: 52-6. https://doi.org/10.37616/2212-5043.1009 PMid:33154892 PMCid:PMC7640594 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

