Association between disease duration, GH-IGF-1 axis markers, and linear growth in children with type 1 diabetes mellitus: a cross-sectional study
ORIGINAL RESEARCH ARTICLE
Association between disease duration, GH-IGF-1 axis markers, and linear growth in children with type 1 diabetes mellitus: a cross-sectional study
Article Summary
- DOI: 10.24969/hvt.2026.642
- CARDIOVASCULAR DISEASES
- Published: 15/04/2026
- Received: 10/02/2026
- Revised: 06/04/2026
- Accepted: 07/04/2026
- Views: 455
- Downloads: 282
- Keywords: Type 1 diabetes mellitus, height, insulin-like growth factor I, growth hormone, HBA1C, disease duration
Address for Correspondence: Feruzakhan Uvaidillaeva, Kyrgyz-Russian Slavic University named after B.N. Yeltsin, Bishkek, Kyrgyz Republic
Email: Feruzuvaidillaeva@gmail.com
ORCID: Feruzakhan Uvaidillaeva: 0000-0003-3657-0083; Rustam Tuhvatshin 0000-0002-9329-8568; Valeriia Kniazeva: 0000-0002-3357-6298; Baktygul Omurkulova:0009-0000-8070-4451 Facebook: feruza.uvaidillaeva
Feruzakhan Uvaidillaeva ¹, Rustam Tuhvatshin 2, Valeriia Kniazeva 3, Baktygul Omurkulova 3
1 Kyrgyz-Russian Slavic University named after B.N. Yeltsin, Bishkek, Kyrgyz Republic
2 Kyrgyz State Medical Academy named after I.K. Akhunbaev, Bishkek, Kyrgyz Republic
3 International Higher School of Medicine, Bishkek, Kyrgyz Republic
Abstract
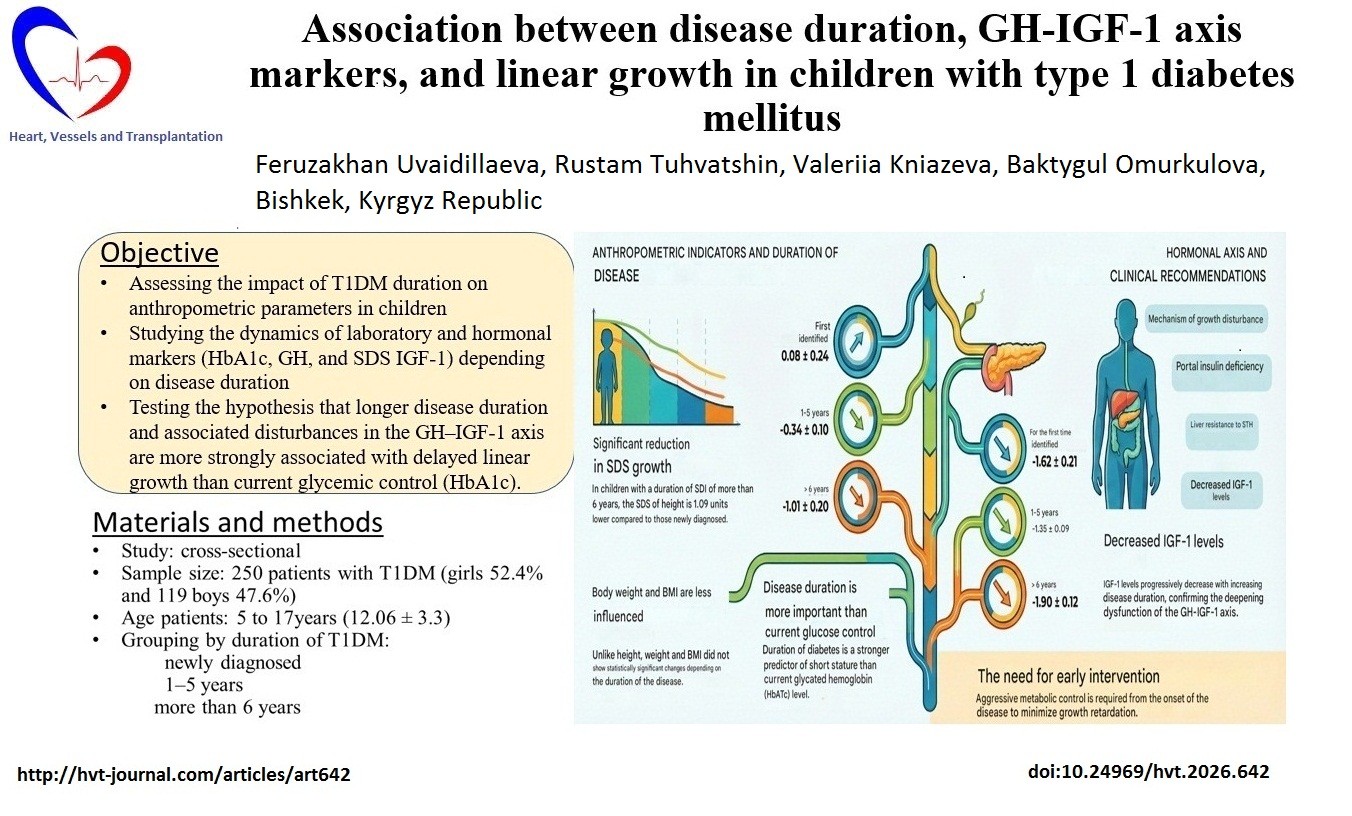
Objective: This single-center cross-sectional study analyzed 250 children and adolescents (aged 5–17 years) with type 1 diabetes mellitus (T1DM) in the Kyrgyz Republic to assess the effect of T1DM duration on anthropometric parameters and key hormonal growth markers (glycated hemoglobin A1c (HbA1c), growth hormone (GH), and insulin-like growth factor 1 (IGF-1) SDS). We hypothesized that longer duration of chronic T1DM and related GH–IGF-1 axis disruption are more strongly associated with linear growth disorders than current glycemic control.
Methods: Anthropometric data (SDS height/weight) and hormonal levels were compared across groups categorized by disease duration (newly diagnosed, 1–5 years, and >6 years).
Results: Children with T1DM duration over 6 years exhibited markedly lower height SDS compared to both newly diagnosed (p<0.001) and 1–5-year groups (p=0.006). Mean IGF-1 SDS significantly decreased with increasing disease duration (p=0.010), indicating worsening GH–IGF-1 axis dysfunction. Although mean HbA1c improved gradually with duration, the overall level remained suboptimal (10.7%). The duration of the disease (β = –0.338; 95%CI -0.204 – -0.097, p < 0.001), the age of T1DM manifestation (β = 0.153; 95%CI 0.011 – 0.105, p = 0.015), SDS IGF1 (β = -0.245; 95% CI 0.046 – 0.254, p = 0.012) and SDS weight (β = 0.524; 95% CI 0.484 – 0.718, p < 0.001) had a significant effect on the SDS of height, while the other GH variables (β = 0.037; p = 0.558), HbA1c (β = –0.015; p = 0.809) did not make a statistically significant contribution.
Conclusions: These findings confirm that longer disease duration is significantly associated with linear growth impairment in children with T1DM, underscoring the need for early and aggressive intervention to minimize cumulative exposure.
Key words: Type 1 diabetes mellitus, height, insulin-like growth factor I, growth hormone, HBA1C, disease duration

Graphical abstract
Introduction
Type 1 diabetes mellitus (T1DM) is one of the most common chronic endocrine diseases affecting children and adolescents (1). Given its high prevalence (2) and potential long-term effects, monitoring children's physical growth and development is vital for their overall care and well-being (1, 3). Adolescents with poor control may experience stunted growth. T1DM, like other chronic illnesses, can impact growth and puberty (4).
Growth issues in T1DM are closely linked to the dysregulation of the growth hormone (GH)–insulin-like growth factor 1 (IGF-1) axis (4, 5). These alterations result from poor metabolic control and endogenous insulin deficiency. Insulin plays a crucial role in this axis, as GH's action must be translated into IGF-1 production by the liver (3). When insulin does not reach the liver via the portal vein, the liver becomes resistant to GH, leading to decreased IGF-1 production and serum levels. In response, the pituitary gland compensatorily secretes more GH, causing hypersecretion of GH. These changes in the GH/IGF-1 axis- lower IGF-1 and higher GH levels- are characteristic features seen in patients with T1DM (5).
Previous studies on how T1 DM affects growth and final height offer mixed results (2). Some suggest that height reduction is most noticeable when disease occurs before puberty, although children might be taller at debut (5). It is widely accepted that poor glycemic control and longer disease duration influence height (6, 7). Nonetheless, despite these observations, the direct link between uncontrolled diabetes, early T1DM diagnosis, and increased risk of disrupted final linear growth remains uncertain, warranting further research.
Since anthropometric data in children with T1DM can be limited in some regions (1), and to better understand the mechanisms behind growth impairment, this study analyzed a large sample of children and adolescents with T1DM.
The goal was to evaluate how T1DM duration affects growth parameters (height, weight, body mass index (BMI)) and to observe changes in HbA1c and key hormonal markers (GH and IGF-1 SDS) based on disease duration.
We hypothesized that longer duration of chronic T1DM and related GH–IGF-1 axis disruption are more strongly associated with linear growth disorders than current glycemic control (glycated hemoglobin A1c (HbA1c)).
Methods
Study design and sampling
A single-center cross-sectional observational study with T1DM was carried out in the endocrinology department of the National Center for Maternal and Child Health of the Kyrgyz Republic from October 2023 to October 2024.
A convenience sampling method was employed, involving 250 children aged 5 to 17 years with a diagnosis of T1DM, who were recruited during their inpatient visits and provided informed consent. Exclusion criteria: age under 5 years, concomitant diseases in children, congenital hypothyroidism, pituitary nanism, refusal to participate, and severe somatic and mental diseases. Patients` groups were stratified by diabetes duration (0, 1–5, and >6 years) to assess the long-term impact of metabolic control on linear growth, which is a recognized methodological approach in studying the chronic complications of diabetes: with newly diagnosed diabetes (n=41), with a diabetes history of 1–5 years (n=151), and with a diabetes history of >6 years (n=58).
Ethical review: the study protocol was approved by the Ethics Committee of the Kyrgyz Medical Academy named after I. K. Akhunbaev, protocol number 18. dated 05/15/2022. All subjects were examined after providing informed consent from their parents/guardians. The study was conducted according to the rules of Helsinki 2024 agreement on human studies.
Baseline variables
We collected demographic data (age, sex), medical history data as the total duration of diabetes and age at onset of T1DM. Clinical examinations included anthropometric measurements, physical examination and laboratory data evaluation. Treatment information: all patients received insulin replacement therapy and were trained in self-management programs.
Anthropometric measurements
Anthropometric measurements included standard measurements of height, body weight, and body mass index (BMI) were calculated. Anthropometric indicators were standardized against age-sex norms and reported as standard deviations (SDS) according to WHO growth standards (11).
Laboratory and hormone analysis
The following laboratory parameters were determined to assess the metabolic status and hormonal profile:
1. HbA1c: Measured to assess mean glycemic control over the previous 3 months.
2. GH and IGF-1 hormone concentrations were determined using the GH and IGF-1 ELISA Kit from DRG International, a Biocheck Company (Germany), using an enzyme-linked immunosorbent assay (ELISA) (Mindray MR-96A). Blood sampling was carried out in the morning, on an empty stomach (at least 8 hours after meals). The GH level was determined in accordance with the standards specified in the assay kit (reference range 0.1–7.7 ng/mL).
3. IGF-1 SDS values were calculated using an online calculator (Child Metrics, Turkish Society for Pediatric Endocrinology and Diabetes) (https://childmetrics.org/IGF), based on age- and sex-specific reference data.
Statistical analysis
Statistical processing of the study results was carried out using the program "SPSS", version 16.0 for Windows (IBM, New York, USA). To assess differences between groups of patients with different durations of T1DM (newly identified, 1–5 years, more than 6 years), appropriate method of comparative analysis for multiple groups (ANOVA) was used, and the results are presented as F and p-values.
The relationships between the variables (duration of T1DM, SDS growth, HbA1c, SDS IGF1, GH) were assessed using correlation analysis (Pearson).
Multiple linear regression analysis was used to determine the independent effect of various clinical and hormonal variables on growth SDS. Variables such as disease duration, age of onset of T1DM, SDS IGF-1, weight SDS, GH, and HbA1c were included in the regression model. Statistically significant differences at the p<0.05 level were considered.
Results
The study included 250 children with T1DM, including 131 (52.4%) girls and 119 (47.6%) boys. The average age of the children was 12.1 (3.3) years. The average HbA1c level was 10.7 (2.7)%.
Comparative analysis showed that the duration of T1DM showed a pronounced effect on anthropometric growth indicators. Children with T1DM for more than 6 years demonstrated a 1.09 unit decrease in mean SDS height compared to those with newly diagnosed T1DM (from 0.08 to -1.01). Although body weight (SDS) and BMI also showed a tendency to change with T1DM duration, these differences did not reach statistical significance (p = 0.070; p = 0.072).
In children newly diagnosed with diabetes, the HbA1c was significantly higher (p < 0.001), reaching 12.3 (2.5)%. In the group of children with T1DM for 1 to 5 years, this indicator decreased to 10.5 (2.7)%, and in children with T1DM for more than 6 years, it was 10.5 (2.6)%. IGF-1 SDS levels in patients with different durations of diabetes mellitus revealed significant differences (p = 0.010). The data demonstrated that mean IGF-1 SDS values decrease with increasing T1DM duration (Table 1).
|
Table 1. Anthropometric and laboratory parameters in children with T1DM depending on the duration of the disease |
||||||
|---|---|---|---|---|---|---|
|
Index |
n=250 |
Newly diagnosed (n=41) |
1–5 years (n=151) |
>6 years (n=58) |
F |
p (ANOVA) |
|
SDS height |
-0.42 (1.4) |
0.08 (1.5) |
-0.34 (1.3) |
-1.01 (1.5) |
8.1 |
<0.001** |
|
SDS weight |
-0.52 (1.3) |
-0.55 (1.5) |
-0.38 (1.2) |
-0.84 (1.3) |
2.7 |
0.070 |
|
BMI, kg/m2 |
17.5 (2.8) |
16.6 (3.1) |
17.6 (2.6) |
17.8 (2.9) |
2.7 |
0.072 |
|
HbA1c, % |
10.8 (2.7) |
12.3 (2.5) |
10.5 (2.7) |
10.5 (2.6) |
7.9 |
<0.001** |
|
GH, ng/ml |
6.9 (1.2) |
3.9 (0.6) |
7.4 (0.9) |
7.7 (1.3) |
2.4 |
0.092 |
|
IGF1 SDS |
-1.5 (1.2) |
-1.62 (1.4) |
-1.35 (1.2) |
-1.90 (0.9) |
4.7 |
0.010* |
|
Data are presented as mean (SD). ANOVA test for independent groups. p<0.01* — statistically significant, p <0.001** - statistically significant BMI – body mass index, GH – growth hormone, IGF 1 - insulin-like growth factor I, T1DM – type I diabetes mellitus |
||||||
Correlation analysis showed that the duration of T1DM is negatively and significantly correlated with the growth of SDS (r = -0.33, p < 0.001), with the level of HbA1c (r = -0.15, p = 0.01), and with SDS IGF1 (r = -0.14, p = 0.02). No statistically significant correlation between HbA1c and growth was found (p = 0.446). Similarly, the association between HbA1c and SDS IGF1 was not statistically significant.
To determine the direct influence of factors on the growth of SDS, a multiple linear regression was performed. The duration of the disease (β = –0.338; p < 0.001), the age of T1DM manifestation (β = 0.153; p = 0.015), SDS IGF1 (β = -0.245; p = 0.012) and SDS weight (β = 0.524; p < 0.001) had a significant effect on the SDS of height, while the other GH variables (β = 0.037; p = 0.558), HbA1c (β = –0.015; p = 0.809) did not make a statistically significant contribution (Table 2).
|
Table 2. Multiple linear regression for SDS height |
|||||
|
Index |
β |
SE |
t |
p |
95% CI |
|
Duration of the disease |
–0.333 |
0.027 |
-5.556 |
<0.001 |
-0.204 – -0.097 |
|
T1DM manifestation |
0.153 |
0.024 |
2.441 |
0.015 |
0.011 – 0.105 |
|
SDS IGF1 |
-0.245 |
0.076 |
1.360 |
0.012 |
0.046 – 0.254 |
|
SDS weight |
0.524 |
0.060 |
10.101 |
<0.001 |
0.484 – 0.718 |
|
HbA1c |
-0.015 |
0.034 |
-0.242 |
0.809 |
-0.074 – 0.058 |
|
CI – confidence interval, HbA1c – glycated hemoglobin A1c, IGF 1 - insulin-like growth factor I, T1DM – type I diabetes mellitus |
|||||
Discussion
In this study, it should be noted that the average HbA1c level in the sample (10.7%) is quite high, which indicates suboptimal or poor glycemic control in a significant proportion of patients, since optimal control, according to international recommendations, is usually defined as a level below 7.0% (8).
Anthropometric disorders and disease duration of T1DM
The main result of the study is that the duration of T1DM is a factor significantly associated with lower linear growth. Comparative analysis showed that children with T1DM duration over 6 years of age showed markedly low SDS growth rates compared to both children with newly diagnosed T1DM (p < 0.001) and children with the disease from 1 to 5 years of age (p = 0.006). This result is consistent with the data of other studies that have found that a longer course of the disease and an earlier age of T1DM manifestation are associated with worse growth and a decrease in final height (5). For example, patients with growth retardation have been shown to have an earlier age of onset and a longer duration of diabetes (4).
The results of the correlation analysis confirm the complex relationships between disease duration, metabolic control, and height. This suggests a potential link between disease duration and stunted growth of chronic disease, which is a powerful factor that negatively affects linear growth. Many studies, including longitudinal studies, have shown that the duration of diabetes is a key factor associated with stunted growth and reduced final height (8).
Interestingly, although body weight (SDS) and BMI also showed a tendency to change with the duration of T1DM, these differences did not reach statistical significance. This may indicate that while the effect of disease duration on linear height is pronounced, the effect on weight is less straightforward in this cohort. However, other studies note that children with T1DM may be shorter and slimmer compared to the control group (1), although in recent years there has been a trend towards an increase in BMI and the prevalence of obesity in children with T1DM against the background of improved insulin therapy (9).
Dynamics of glycemic control (HbA1c)
The HbA1c analysis showed that the highest rate was observed in children with newly diagnosed diabetes. This is expected, since during the period of manifestation of the disease, metabolic control is absent or extremely insufficient. In the groups with the duration of the disease from 1 to 5 years and more than 6 years, this indicator decreased. These data suggest a gradual improvement in HbA1c control with increasing time from disease onset, reflecting the initiation of treatment and education programs, which is supported by a statistically significant, albeit weak, negative correlation between the duration of T1DM and HbA1c levels. This reflects the initiation and intensification of treatment (9)
However, despite the formal improvement over the onset, the mean HbA1c level remains in the category of poor glycemic control (above 9.0%). It should be noted that the lack of a statistically significant association with current HbA1c levels in the regression model (p = 0.809) does not mean that glycemic control is not important for growth. A large Swedish study found that poor glycemic control (HbA1c > 9%) was associated with a significant reduction in final height and an increased risk of short stature in boys. Thus, even if the control is improved, prolonged exposure to high HbA1c levels (as in the >6 years group) is the main factor negatively affecting growth (8).
Hormonal indicators and the GH-IGF-1 axis
Hormonal results show significant differences in SDS IGF-1 levels depending on the duration of T1DM (p = 0.010). At the same time, the mean values of IGF-1 SDS show a decrease depending on the duration of T1DM. The decrease in IGF-1 levels with increasing duration of the disease is a critical finding, as it indicates an aggravation of GH-IGF-1 axis dysfunction (5, 3). This is confirmed by the negative correlation between the duration of T1DM and SDS IGF1. This decrease in IGF-1, which is a key mediator of growth, explains the observed disruption of linear growth (10).
In conditions of chronic lack of portal insulin, liver resistance to GH develops, which leads to a decrease in the production of IGF-1. This decrease in IGF-1 levels is directly related to impaired linear growth (4). The fact that IGF-1 SDS decreases with increasing duration of T1DM, despite the tendency to improve HbA1c, may indicate that compensation for systemic hyperglycemia by subcutaneous insulin administration is not able to fully restore the normal function of the GH-IGF-1 axis, since it does not eliminate insulin deficiency in the portal vein (10). This leads to continued growth disorders, which is confirmed by data on low growth rates in the group with a long history of diabetes (1).
At the same time, there was a tendency for the GH level to gradually increase from the minimum to the maximum corresponding to the norm as the duration of T1DM increased, although this increase did not reach statistical significance (p > 0.05). This is consistent with a pathophysiological model in which a decrease in IGF-1 leads to compensatory hypersecretion of GH due to a weakening of the negative feedback mechanism (5). It must be explicitly stated that single-point fasting GH measurement is not a reliable method for assessing the secretory status of the GH-IGF-1 axis in this study. Due to the distinct pulsatile pattern of GH, these results should not be used to draw definitive physiological conclusions regarding the hypersecretion or deficiency of the hormone.
Dynamic testing or profiling is necessary to make definitive conclusions about GH levels. This limitation likely explains why the observed trend toward increased GH did not reach statistical significance in the group comparison (p=0.092). For future studies aiming at a more detailed evaluation of liver sensitivity to growth hormone, it is recommended to use more stable indicators. Specifically, calculating the IGF-1/GH ratio can act as an additional research marker for GH-IGF-1 axis dysfunction, as it reflects the liver's sensitivity to GH and encompasses both hypersecretion of GH and reduced IGF-1 production.
To better understand the relationship between clinical and hormonal parameters, as well as anthropometric indicators, correlation and multiple linear regression analyses were conducted.
Independent predictors of growth and the role of chronic exposure
The most significant independent predictors of SDS growth were:
1. Disease duration. The fact that disease duration was the strongest factor inversely associated with growth performance confirms the significance of chronic metabolic imbalance over many years. It is a clinical variable reflecting the chronological progression of the disease. This explains why the current HbA1c level, which reflects carbohydrate metabolism status only over the past three months, did not show a statistically significant independent association with the SDS growth score within this regression model (8).
2. Weight SDS, which reflects the expected relationship, since height and weight are interrelated anthropometric indicators; taller children tend to have more weight (4, 10).
3. Age of T1DM manifestation. A positive β ratio indicates that a later age of onset is associated with better growth rates. This is consistent with evidence that the onset of diabetes before puberty can lead to lower final height (5).
4. IGF1 SDS. Although physiologically IGF-1 stimulates growth, its negative β coefficient in this model may indicate complex dysfunction of the GH-IGF-1 axis in a poorly controlled cohort, where a decrease in IGF-1 is part of a pathological process leading to growth retardation (8). The key clinical finding of the regression analysis is that HbA1c and GH did not make a statistically significant independent contribution to growth SDS in this multiple model.
Study limitations
Considering the methodological limitations, we view the GH data exclusively as an auxiliary research marker with low diagnostic reliability for axis evaluation in this cross-sectional setting. Consequently, our conclusions regarding the GH-IGF-1 axis are primarily based on IGF-1 SDS levels, which provide a more stable and reliable reflection of the axis's functional state in children with T1DM. In our regression model, the lack of a statistically significant independent association between the current HbA1c level and height SDS (p = 0.809) should be interpreted with caution. This finding suggests that a single measurement of HbA1c, reflecting glycemic control only over the preceding three months, may not capture the long-term physiological impact as effectively as disease duration. In this specific model, disease duration likely serves as a proxy for cumulative metabolic exposure, which showed a stronger association with linear growth impairment. While extensive longitudinal research has established that chronic poor glycemic control (HbA1c > 9.0%) is associated with significantly lower final height, the cross-sectional design of our study and the absence of historical longitudinal data are primary limitations (8). Without these historical records, we were unable to calculate the cumulative glycemic load throughout the entire disease period. Therefore, the observed links between disease duration, IGF-1 levels, and growth should be viewed as significant associations rather than definitive causal relationships, and future longitudinal studies are essential to confirm these temporal dynamics.
Conclusion
Our data confirm that children with T1DM have impaired linear growth, which is closely linked to duration of the disease and, as a result, the violation of the IGF-1 axis. Although glycemic control improves over time, it remains suboptimal, and the prolonged disease duration is strongly associated with growth retardation. This is especially relevant since abnormalities in the GH/IGF-1 axis in patients with T1DM due to low port
The clinical implications of these findings are the need for early and aggressive intervention to achieve optimal metabolic control, especially in children with early onset of T1DM. Regular monitoring of SDS for height, weight, and IGF-1 SDS should be a mandatory component of assessing the long-term prognosis in this patient population.
al insulin levels may not fully be normalized with systemic insulin administration, even with improved glycemia.
Ethics: All subjects were examined after providing informed consent from their parents/guardians. The study protocol was approved by the Ethics Committee of the Kyrgyz Medical Academy named after I. K. Akhunbaev, protocol number 18. dated 05/15/2022. The study was conducted according to the rules of Helsinki 2024 agreement on human studies.
Peer-review: External and internal
Conflict of interest: None to declare
Authorship: F.U. - study concept and design, data collection and processing, statistical data processing, manuscript writing; R.T.– study concept and design, scientific editing, and approval of the final version of the manuscript;
V.K.– data collection, analysis, and technical processing of primary data; B.O.– data collection, analysis, and participation in manuscript preparation. All authors approved the final version of the article before publication and agreed to be accountable for all aspects of the work, including appropriately investigating and resolving questions related to the accuracy or integrity of any part of the work. Thus equally contributed to the study, preparation of manuscript and fulfilled all authorship criteria
Acknowledgements and funding: None to declare
Statement on A.I.-assisted technologies use: Artificial Intelligence was used only for the creation of the graphical abstract to provide a visual representation of the study’s pathophysiological model. No AI tools were used for data analysis or the writing of the manuscript text.
Data and material availability: The data are available from the corresponding author upon reasonable request
References
| 1.Mynepally M, Yadav BR, Mythili A, Vivekananda1B, Kandregula A. V. Subrahmanyam. Study of growth parameters in children and adolescents with type 1 diabetes mellitus. J Pediatr Endocrinol Diabetes 2023; 3: 58-62. https://doi.org/10.25259/JPED_36_2022 |
||||
| 2.Hovsepian S, Chegini R, Alinia T, Ghaheh HS, Nouri R, Hashemipour M. Final height in children and adolescents with type 1 diabetes mellitus: A systematic review and meta-analysis. Hormones 2024; 23: 35-48. https://doi.org/10.1007/s42000-023-00500-3 PMid:37914868 |
||||
| 3.Nijenhuis-Noort EC, Berk KA, Neggers SJCMM, Lely AJV. The fascinating interplay between growth hormone, insulin-like growth factor-1, and Insulin. Endocrinol Metab (Seoul) 2024; 39: 83-9. https://doi.org/10.3803/EnM.2024.101 PMid:38192102 PMCid:PMC10901670 |
||||
| 4.Thabet RA, Sherif EM, ElAal AOA, Mahmoud RA. Insulin-like growth factor 1 and sex hormones for assessment of anthropometric and pubertal growth of Egyptian children and adolescents with type 1 diabetes mellitus (single-center study). BMC Endocr Disord 2024; 62. https://doi.org/10.1186/s12902-024-01596-3 PMid:38724932 PMCid:PMC11080226 |
||||
| 5.Jadhav I, Chakole S. Effects of type 1 diabetes mellitus on linear growth. A comprehensive review. Cureus 2023; 15. https://doi.org/10.7759/cureus.45428 |
||||
| 6.Koren D. Growth and development in type 1 diabetes. Curr Opinion Endocr Diabet Obes 2022; 29: 57-64. https://doi.org/10.1097/MED.0000000000000694 PMid:34864760 |
||||
| 7.Salih FT, Abdullah WH, Ibrahim BA, Ayoub NI. Assessment of growth status in children and adolescents with type 1 diabetes mellitus in Baghdad a case-control study. JPMA 2024; 74: 48-51. https://doi.org/10.47391/JPMA-BAGH-16-12 PMid:39434271 |
||||
| 8.Smew AI, Lundholm C, Gong T, Lichtenstein P, Sävendahl L, Almqvist C. Glycemic control and adult height: A nationwide Swedish cohort study on childhood type 1 diabetes. J Clin Endocrinol Metab 2025; 110: 2765-77. https://doi.org/10.1210/clinem/dgae809 PMid:39556501 PMCid:PMC12261078 |
||||
| 9.Matsuura N, Yokota Y, Kazahari K, Sasaki N, Amemiya S, Ito Y, et.al., Japanese study group of insulin therapy for childhood and adolescent diabetes. Pediatr Diabetes 2001; 2: 160-9. https://doi.org/10.1034/j.1399-5448.2001.20404.x PMid:15016181 |
||||
| 10.Saare L, Peet A, Tillmann V. Growth in children with HLA-conferred susceptibility to type 1 diabetes. Endocrinol Metab (Seoul) 2022; 37: 175-9. doi 10.3803EnM.2021.1262 https://doi.org/10.3803/EnM.2021.1262 PMid:35255609 PMCid:PMC8901960 |
||||
| 11.https://www.who.int/tools/child-growth-standards/standards | ||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

