Treatment of a ruptured aorto-iliac aneurysm with right iliac arteriovenous fistula: Surgical considerations in the endovascular era
CASE REPORT
Treatment of a ruptured aorto-iliac aneurysm with right iliac arteriovenous fistula: Surgical considerations in the endovascular era
Article Summary
- DOI: 10.24969/hvt.2026.647
- CARDIOVASCULAR DISEASES
- Published: 01/05/2026
- Received: 17/02/2026
- Revised: 24/05/2026
- Accepted: 24/05/2026
- Views: 44
- Downloads: 27
- Keywords: Endovascular aneurysm repair, aorto-caval fistula, iliac arteriovenous fistula, ruptured abdominal aortic aneurysm, open surgical repair, dyspnea, heart failure
Address for Correspondence: Fabio Massimo Oddi, Vascular Surgery Unit, University of Rome “Tor Vergata”, Rome, Italy
Email: Fabio.massimo89@gmail.com
ORCID: Fabio Massimo Oddi - 0000-0001-8081-807X; Andrea Cuoghi -0009-0002-6721-5017; Grazia Granata -0009-0000-5452-652X; Federico Francisco Pennetta – 0000-0002-2477-6652; Eugenio Martelli – 0000-0001-5764-0082
Fabio Massimo Oddi*, Andrea Cuoghi, Gianluca Citoni, Adelaide Borlizzi, Grazia Granata, Manuel Romano, Federico Francisco Pennetta, Eugenio Martelli, Alessandro Ranucci
Vascular Surgery Unit, University of Rome “Tor Vergata”, Rome, Italy
Abstract
Objective: Rupture of abdominal aortic aneurysms (AAA) into the iliac or caval veins through arteriovenous fistulas (AVFs) is a rare but life-threatening complication. We report a case of a 74-year-old man with complex aorto-iliac aneurysmal disease and a spontaneous right iliac AVF, presenting with worsening dyspnea and signs of congestive heart failure.
Case presentation: A 74-year-old man with complex aorto-iliac aneurysmal disease and a spontaneous right iliac AVF, presented with worsening dyspnea and signs of congestive heart failure. Imaging revealed a 45 mm infrarenal AAA, a 50 mm right common iliac artery aneurysm, and an 87 mm right external iliac artery aneurysm. The anatomical configuration—including a short, angulated proximal neck and severely tortuous iliac arteries—rendered the patient unsuitable for endovascular repair. Therefore, an emergent open surgical repair was performed, consisting of aneurysmectomy, AVF closure, and aorto-bi-iliac reconstruction using a Dacron graft. Postoperative recovery was uneventful, with improvement in cardiac and renal function.
Conclusion: This case highlights the ongoing relevance of open surgical approaches in an era dominated by endovascular techniques, particularly when anatomical or hemodynamic challenges limit the feasibility of minimally invasive options.
Key words: Endovascular aneurysm repair, aorto-caval fistula, iliac arteriovenous fistula, ruptured abdominal aortic aneurysm, open surgical repair, dyspnea, heart failure

Introduction
Spontaneous aorto-caval or iliac arteriovenous fistulas (AVFs) represent rare but serious complications of abdominal aortic aneurysms. First described by Syme in 1831 (1) and later systematically studied by Matas (2), these fistulas have historically been treated with open surgical repair. In recent decades, the advent of endovascular aneurysm repair (EVAR) has revolutionized the management of aneurysmal disease.
Graphical abstract
Since the first endovascular treatment of a major AVF by Zajko in 1995 (3), several reports have highlighted the feasibility and advantages of minimally invasive techniques (4).
However, despite the growing use of EVAR, certain anatomical configurations and clinical scenarios still mandate open surgery. This case exemplifies such a situation. It provides an opportunity to reflect on the limitations of endovascular techniques and the continued importance of open surgical expertise in vascular surgery.
We report a case of a 74-year-old man with complex aorto-iliac aneurysmal disease and a spontaneous right iliac AVF, presenting with worsening dyspnea and signs of congestive heart failure.
Case report
A 74-year-old male with a history of hypertension, dyslipidemia, and coronary artery disease (status post quadruple bypass) presented to the emergency department with progressive dyspnea (NYHA class III), hematuria, and lower limb edema. He also had a remote history of left nephrectomy.
On examination, the patient had a pulsatile abdominal mass, a continuous machinery-like abdominal bruit, and signs of congestive heart failure. Laboratory results indicated anemia (hemoglobin 11.2 g/dL), leukocytosis (white blood cells count 14,600/mm3), renal impairment (creatinine 2.35 mg/dL, estimated glomerular filtration rate 27 mL/min), and elevated troponin levels (12 ng/L).
Computed tomography angiography revealed a 45 mm infrarenal AAA with a pseudoaneurysm near the left renal artery stump, a 50 mm right common iliac artery aneurysm, and an 87 mm right external iliac artery aneurysm involving the origin of the internal iliac artery (25 mm). Early contrast opacification of the right iliac vein and inferior vena cava confirmed a large iliac AVF (Fig. 1). The proximal neck was short and angulated, and iliac arteries were tortuous—features that contraindicated EVAR.
An emergent open repair via xipho-pubic laparotomy was undertaken. Findings included ascites, venous congestion, and absence of the left renal vein. Right renal artery and ureter were carefully preserved. Aortic clamping was performed under close anesthesiological monitoring to avoid hemodynamic collapse. The aneurysm sac was opened, and bleeding from the AVF was controlled with digital compression and gauze tamponade.
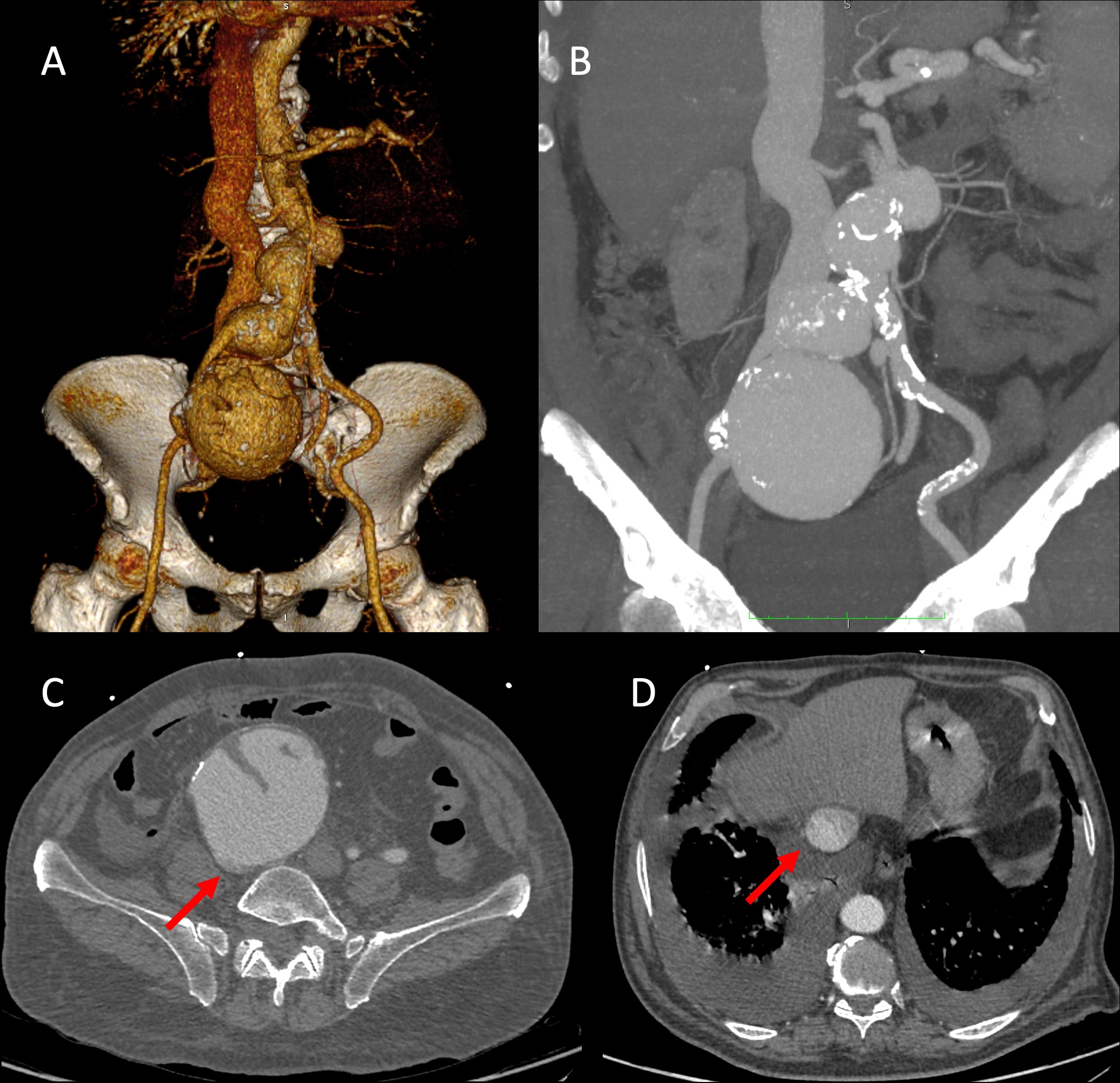
Figure 1. CTA documented infrarenal AAA, a right common iliac artery aneurysm, and a right external iliac artery aneurysm involving the origin of the internal iliac artery (A - B). The right iliac artery aneurysm presses the ipsilateral iliac vein (C). Same contrast opacification of the inferior vena cava with pleural effusion confirmed AVF with heart failure (D).
AAA – abdominal aortic aneurysm, AVR – arterio-venous fistula, CTA- computed tomography angiography
The fistula was sutured with Prolene 3-0, and an 18x9 mm aorto-bi-iliac Dacron graft was anastomosed. The right external iliac artery was revascularized, and pelvic perfusion was ensured through the contralateral hypogastric artery.
The postoperative course was favorable. The patient was extubated in ICU after 4 days and discharged on postoperative day 10. Renal function normalized, hematuria resolved, and cardiac function improved. He was briefly readmitted one week later for community-acquired pneumonia, which resolved with antibiotics.
Discussion
Iliocaval AVFs complicate 3–6% of ruptured abdominal aortic aneurysms (5, 6). Though rare, their hemodynamic impact is profound. A high-flow shunt from artery to vein increases preload, reduces systemic vascular resistance, and often leads to high-output cardiac failure (7, 8). Renal impairment is common due to both arterial hypoperfusion and venous congestion (9).
The classic triad—abdominal pain, pulsatile mass, and bruit—is only present in a minority of patients (8). More often, symptoms are nonspecific and attributed to congestive heart failure or renal dysfunction. In our case, hematuria and heart failure were key presenting features (10, 11).
Surgical management is challenging. Open repair requires precise clamping and delicate fistula closure to prevent air or thrombotic embolism (6). The presence of thrombus in the aneurysm sac adds to the embolic risk (12). Gradual aortic clamping and assistant-applied compression of the AVF are vital techniques to reduce intraoperative bleeding and maintain hemodynamic stability.
Endovascular repair, although it appears better due to its reduced invasiveness and blood loss, is not always feasible (4). Hostile neck anatomy, severe tortuosity, and risk of persistent endoleaks or unsealed AVFs or other complications limit its application (13).
Moreover, long-term data comparing open and endovascular repair for AVF remain scarce and inconclusive (14, 15).
Recent meta-analyses show mixed outcomes, with similar mortality but higher complication rates in some endovascular cohorts.
This case underscores that open surgery retains a critical role in vascular practice. As the vascular community increasingly embraces EVAR, the technical and decision-making skills required for open repair must not be lost. Dedicated training and careful patient selection remain paramount.
Conclusion
In the endovascular era, open surgical repair continues to offer definitive treatment for patients with complex aorto-venous fistulas and unsuitable anatomy for EVAR. Despite advances in stent-graft technology, anatomical and hemodynamic considerations still dictate individualized treatment strategies. The case illustrates that open surgery, when properly indicated and executed, remains a life-saving and effective solution. A multidisciplinary approach and surgeon expertise are essential for optimal outcomes.
Ethics: Informed consent was obtained for publication of this case report and follow-up imaging. The study was performed in frame of Helsinki 2024 agreement for human studies.
Peer-review: External and Internal
Conflict of interest: None to declare
Authorship: F.M.O., A.C., G.C., A.B., G.G., M.R., F.F.P., E.M., A.R. equally contributed to the management of the patients and preparation of case report, thus fulfilled all authorship criteria
Acknowledgements: The authors thank the surgical and ICU teams involved in the patient's care
Funding: None to declare
Statement on A.I.-assisted technologies use: Authors used artificial intelligence (A.I.) tools for preparation of graphical abstract
Data and material availability: Does not apply
References
| 1.Syme J. Case of spontaneous varicose aneurysm. Edimb Med J 1831; 36: 104-5. | ||||
| 2.Matas R, Keen WW. Surgery: Its Principles and Practice. Philadelphia 1909; 5: 293. | ||||
| 3.Zajko AB, Little AF, Steed DL, Curtiss EI. Endovascular stent-graft repair of common iliac artery-to-inferior vena cava fistula. J Vasc Interv Radiol 1995; 6: 803-6. doi: 10.1016/s1051-0443(95)71190-3 https://doi.org/10.1016/S1051-0443(95)71190-3 PMid:8541688 |
||||
| 4.Antoniou GA, Koutsias S, Karathanos C, Sfyroeras GS, Vretzakis G, Giannoukas AD. Endovascular stent-graft repair of major abdominal arteriovenous fistula: a systematic review. J Endovasc Ther 2009; 16: 514-23. doi: 10.1583/09-2725.1 https://doi.org/10.1583/09-2725.1 PMid:19702345 |
||||
| 5.Fenster MS, Dent JM, Tribble C, Angle JF, Sarembock IJ, Komada M, et al. Aortocaval fistula complicating abdominal aortic aneurysm: case report and literature review. Cathet Cardiovasc Diagn 1996; 38: 75-9. doi: 10.1002/(SICI)1097-0304(199605)38 https://doi.org/10.1002/(SICI)1097-0304(199605)38:1<75::AID-CCD17>3.0.CO;2-R |
||||
| 6.Nakazawa S, Mohara J, Takahashi T, Koike N, Takeyoshi I. Aortocaval fistula associated with ruptured abdominal aortic aneurysm. Ann Vasc Surg 2014 https://doi.org/10.1016/j.avsg.2014.03.015 PMid:24698772 |
||||
| 28: 1793.e5-9. doi: 10.1016/j.avsg.2014.03.015 https://doi.org/10.1016/j.avsg.2014.03.015 PMid:24698772 |
||||
| 7.Reckless JP, McColl I, Taylor GW. Aorto-caval fistulae: an uncommon complication of abdominal aortic aneurysms. Br J Surg 1972; 59: 461-2. doi: 10.1002/bjs.1800590614 https://doi.org/10.1002/bjs.1800590614 PMid:5031192 |
||||
| 8.McDonald GR, Graham KJ, Barratt-Boyes BG. Aortocaval fistulae: an occasional cause of congestive cardiac failure. Aust N Z J Surg 1982; 52: 573-5. doi: 10.1111/j.1445-2197.1982.tb06114.x https://doi.org/10.1111/j.1445-2197.1982.tb06114.x PMid:6962726 |
||||
| 9.Brewster DC, Cambria RP, Moncure AC, Darling RC, LaMuraglia GM, Geller SC, et al. Aortocaval and iliac arteriovenous fistulas: recognition and treatment. J Vasc Surg 1991; 13: 253-64. https://doi.org/10.1016/0741-5214(91)90218-J https://doi.org/10.1067/mva.1991.25811 PMid:1990167 |
||||
| 10.Salo JA, Verkkala KA, Ala-Kulju KV, Heikkinen LO, Luosto RV. Hematuria is an indication of rupture of an abdominal aortic aneurysm into the vena cava. J Vasc Surg 1990; 12: 41-4. https://doi.org/10.1016/0741-5214(90)90367-J https://doi.org/10.1067/mva.1990.19860 PMid:2374253 |
||||
| 11.Culp O, Bernatz PE. Urologic aspects of lesions in the abdominal aorta. J Urol 1961; 86: 189-95. doi: 10.1016/S0022-5347(17)65137-1 https://doi.org/10.1016/S0022-5347(17)65137-1 PMid:13718814 |
||||
| 12.Cooperman M, Deal KF, Wooley CF, Evans WE. Spontaneous aortocaval fistula with paradoxical pulmonary embolization. Am J Surg 1977; 134: 647-9. doi: 10.1016/0002-9610(77)90455-x https://doi.org/10.1016/0002-9610(77)90455-X PMid:920898 |
||||
| 13.Ascoli Marchetti A, Oddi FM, Diotallevi N, Battistini M, Ippoliti A. An unusual complication after endovascular aneurysm repair for giant abdominal aortic aneurysm with aortocaval fistula: High bilirubin levels. SAGE Open Med Case Rep 2020; 25: 2050313X20984322. doi: 10.1177/2050313X20984322 https://doi.org/10.1177/2050313X20984322 PMid:33489236 PMCid:PMC7768567 |
||||
| 14.Orion KC, Beaulieu RJ, Black JH 3rd. Aortocaval fistula: Is endovascular repair the preferred solution? Ann Vasc Surg 2016; 31: 221-8. doi: 10.1016/j.avsg.2015.09.006 https://doi.org/10.1016/j.avsg.2015.09.006 PMid:26597238 PMCid:PMC4860718 |
||||
| 15.Borghese O, Pisani A, Sbenaglia G, Giudice R. Open surgery and endovascular techniques in treatment of acute abdominal arteriovenous fistulas. Ann Vasc Surg 2019; 61: 427-33. doi: 10.1016/j.avsg.2019.03.041 https://doi.org/10.1016/j.avsg.2019.03.041 PMid:31207397 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

