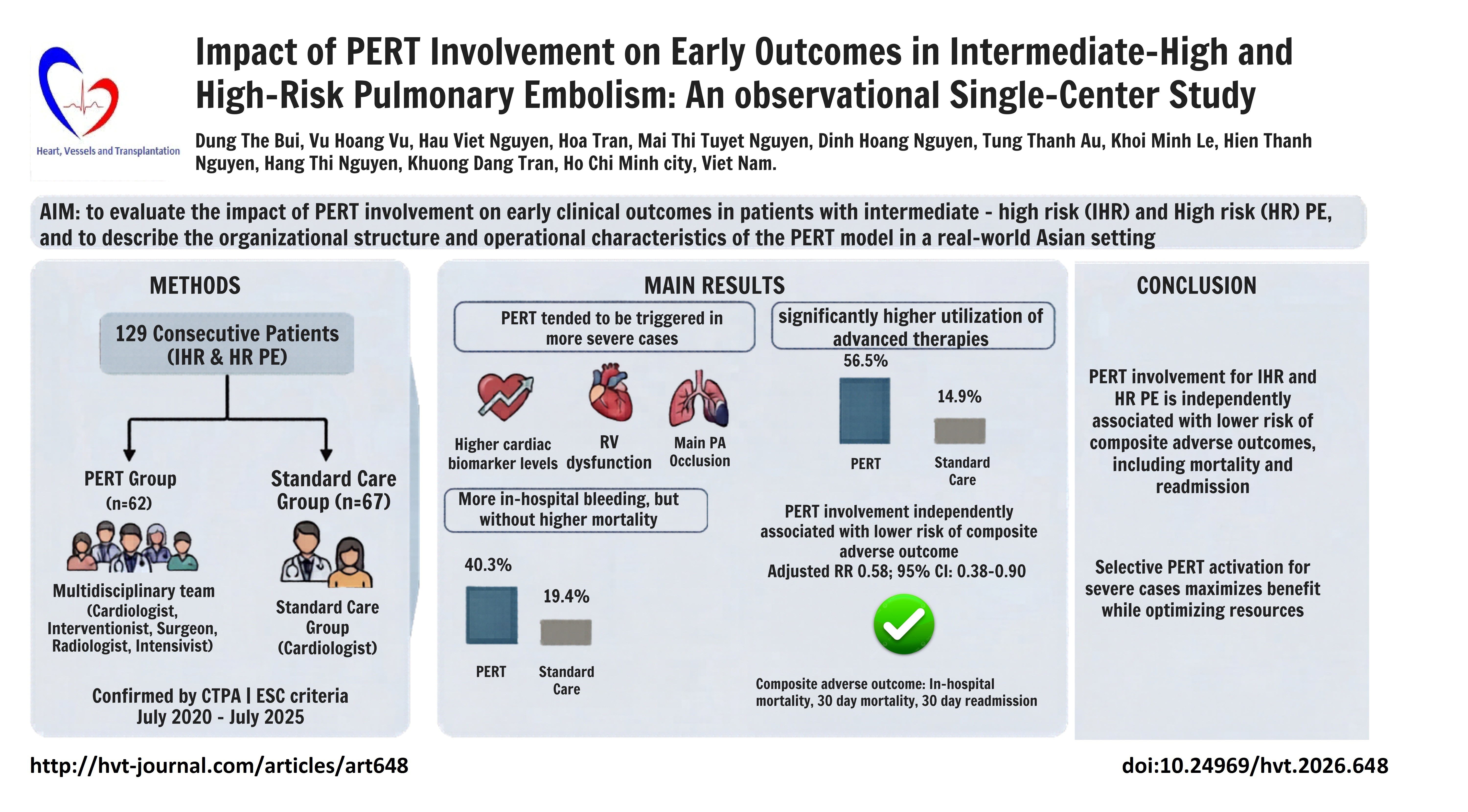
Impact of pulmonary embolism response team involvement on early outcomes in intermediate–high and high-risk pulmonary embolism: An observational single-center study
ORIGINAL RESEARCH ARTICLE
Impact of pulmonary embolism response team involvement on early outcomes in intermediate–high and high-risk pulmonary embolism: An observational single-center study
Article Summary
- DOI: 10.24969/hvt.2026.648
- CARDIOVASCULAR DISEASES
- Published: 01/05/2026
- Received: 10/03/2026
- Revised: 15/04/2026
- Accepted: 16/04/2026
- Views: 128
- Downloads: 33
- Keywords: PERT, pulmonary embolism, composite outcome, high risk, prognosis
Address for Correspondence: Khuong Dang Tran, Apartment C06.07, The Privia Khang Dien Residence, 321 An Duong Vuong Street, An Lac Ward, Binh Tan District, Ho Chi Minh City, Vietnam
Email: khuong.td2@umc.edu.vn Mobile: +84 0352731868
ORCID: Khuong Dang Tran - 0009-0003-7709-9088; Dung The Bui- 0009-0003-8383-5176; Dinh Hoang Nguyen -0000-0002-6769-0849; Vu Hoang Vu - 0000-0001-5482-859X; Hoa Tran - 0000-0001-8393-6442; Hang Thi Nguyen - 0009-0007-7198-099X; Mai Thi Tuyet Nguyen - 0000-0002-1849-9554; Khoi Minh Le - 0000‑0003‑2250‑0818; Hien Thanh Nguyen - 0009-0004-3844-6937; Tung Thanh Au - 0009-0003-9158-3888; Hau Viet Nguyen - 0000-0002-9731-8685
Dung The Bui1a, Vu Hoang Vu2, Hau Viet Nguyen1b, Hoa Tran2, Mai Thi Tuyet Nguyen1a, Dinh Hoang Nguyen1c, Tung Thanh Au2, Khoi Minh Le1d, Hien Thanh Nguyen1a, Hang Thi Nguyen1e, Khuong Dang Tran1a*
1aDepartment of Cardiology, 1bDepartment of Emergency Medicine, 1cDepartment of Heart Surgery, 1dCardiovascular Imaging Unit and 1eDepartment of Radiology, University Medical Center HCMC, Ho Chi Minh City, Vietnam
2Department of Internal Medicine, Faculty of Medicine, University of Medicine and Pharmacy at Ho Chi Minh City, Ho Chi Minh City, Vietnam
Abstract
Objective: Pulmonary embolism response teams (PERTs) facilitate rapid multidisciplinary management of acute pulmonary embolism (PE), particularly in intermediate–high (IHR) and high-risk (HR) patients. However, benefits remain inconsistent, due to PERT structure differences across institutions worldwide.
This study evaluated PERT effectiveness in an Asian healthcare setting, focusing on IHR and HR PE, thereby contributing to the global evidence on PERT.
Methods: This prospective, single-center observational study included 129 consecutive patients with IHR and HR acute PE confirmed by computed tomography pulmonary angiography (CTPA). Patients were managed with or without PERT involvement. The primary outcome was a composite adverse outcome, defined as in-hospital mortality, 30-day post-discharge mortality, or 30-day readmission. Relative risks were estimated using log-binomial regression.
Results: PERT activation tended to occur in patients with higher cardiac biomarker levels and more marked right ventricular dysfunction, patients with cancer were more frequently managed without PERT involvement. Advanced therapy use was higher in the PERT group (14.9% vs. 56.5%, p < 0.001). After adjustment for PESI score, ESC risk category, and discharge anticoagulation status, PERT involvement was independently associated with lower risk of the composite adverse outcome (adjusted RR 0.58, 95% CI 0.38–0.90, p = 0.029).
Conclusions: In patients with IHR and HR acute PE, PERT involvement was independently associated with lower risk of a composite adverse outcome, including mortality and readmission. Selective PERT activation in patients with more severe clinical profiles may maximize benefit while minimizing workload.
Key words: PERT, pulmonary embolism, composite outcome, high risk, prognosis
Introduction
Acute pulmonary embolism (PE) is a common and potentially life-threatening condition, and its management depends on risk stratification into high-risk (HR), intermediate-high risk (IHR), intermediate-low risk or low-risk categories.
Although therapeutic anticoagulation remains the standard initial treatment for IHR PE, the main area of uncertainty is when and in whom to escalate beyond anticoagulation (systemic thrombolysis (ST), catheter-directed therapy, or surgery) before overt hemodynamic collapse. Contemporary recommendations differ across societies.

Graphical abstract
The European Society of Cardiology (ESC) strategy is relatively conservative, emphasizing anticoagulation and close monitoring in IHR PE and reserving reperfusion strategies primarily for hemodynamic deterioration (with catheter-directed or surgical options particularly when ST is contraindicated or has failed) (1). In contrast, CHEST similarly recommends against routine ST in PE without hypotension, but provides explicit “deterioration” features (e.g., falling systolic blood pressure, worsening gas exchange or perfusion, worsening right ventricle (RV) function, rising biomarkers) that may prompt escalation in selected patients prior to frank shock, and suggests catheter-directed thrombectomy (CDTh) mainly in hypotensive patients with high bleeding risk, failed thrombolysis, or impending death (2). This heterogeneity in escalation thresholds and modality selection across guidelines supports the role of multidisciplinary pulmonary embolism response team (PERT) decision-making for IHR PE, where individualized assessment of clinical trajectory, bleeding risk, and local expertise is critical. Moreover, in term of HR patients, Although ST is recommended as first-line therapy, real-world practice is often complicated by contraindications, bleeding risk, advanced age, or comorbid conditions. In such above scenarios, alternative interventions—including catheter-directed therapies, surgical embolectomy — may represent important treatment options. To address the therapeutic uncertainty, many hospitals have established PERT.
PERT is a multidisciplinary rapid-response team (typically including specialists in cardiac surgery, pulmonology, cardiology, hematology, critical care, and radiology) that is available 24/7 and convenes rapidly to evaluate patients with significant PE and coordinate an individualized treatment plan.
Beyond selecting the optimal therapy, PERT members facilitate the fastest possible access to advanced interventions when indicated, provide ongoing in-hospital follow-up, and continue post-discharge surveillance, with re-activation triggered if the patient’s clinical status deteriorates.
Another point to emphasize is that the impact of PERT can vary widely between centers due to differences in practice patterns and team composition. PERT programs differ in which specialists are involved and how aggressively they utilize advanced therapies. For example, a survey of PERT programs found most teams include 3–5 specialists (often from critical care, cardiology, emergency medicine, etc.), but some teams involve up to 10 specialists, and the mix of disciplines varies by institution (3, 4). Likewise, the use of invasive interventions differs markedly: in some hospitals catheter-directed thrombolysis (CDT) is used in 0% of PEs, whereas in others it’s used in ~20% of cases, and overall “advanced therapy” utilization ranges from 16% to 46% of PE patients depending on the center. These disparities likely reflect differences in local expertise, resource availability, and clinical philosophy across PERT programs. As a result, the outcomes and benefits of PERT reported in the literature have shown heterogeneity. Several single-center studies have demonstrated favorable clinical outcomes following the implementation of PERT. A large retrospective cohort study conducted at the Cleveland Clinic reported that the availability of PERT was associated with a significant reduction in 30-day or in-hospital mortality (8.5% vs. 4.7%, p=0.03), with the greatest benefit observed among patients with IHR and HR PE (10.0% vs. 5.3%, p=0.02) (5). However, a meta-analysis including 22 original studies reported that, compared with the pre-PERT era, PERT implementation was not associated with survival benefit even within the subgroup of IHR and HR PE . Importantly, heterogeneity among studies was substantial (6).
While our group previously reported the impact of PERT establishment at our institution, the current study addresses a different research question. Our previous study evaluated the impact of PERT establishment using a before–after design. However, such a design is subject to temporal bias, as improvements in clinical expertise, procedural techniques over time may influence outcomes. In contrast, the current study compares patients managed with and without PERT activation within the same study period, thereby minimizing confounding related to temporal changes. Furthermore, the focus of the present study is restricted to patients with IHR and HR PE, a subgroup that has been shown to derive the greatest potential benefit from multidisciplinary PERT management. Our prior study included patients across all PE risk categories, which may have diluted the effect of PERT (7).
Given this potential variability, it is important to evaluate the performance of PERT in each local context. To date, most PERT studies have been conducted in Europe and North America, whereas data from Asian countries remain limited. Differences in patient characteristics, healthcare infrastructure across Asian centers may substantially influence PERT outcomes. Moreover, evidence specifically focused on patients with IHR and HR PE remains scarce. Therefore, this study aimed to evaluate the impact of PERT involvement on early clinical outcomes in patients with IHR and HR PE, and to describe the organizational structure and operational characteristics of the PERT model in a real-world Asian setting.
Methods
Study design and population
This prospective, single-center observational study enrolled consecutive eligible patients at the University Medical Center Ho Chi Minh City, Vietnam, between July 2020 and July 2025. Inclusion criteria were adult patients (aged ≥18 years) with acute PE confirmed by computed tomography pulmonary angiography (CTPA) who were classified as IHR or HR PE according to the 2019 ESC guidelines. Patients were excluded if they had chronic PE, were already receiving anticoagulation at the time of diagnosis, or were classified as low-risk or intermediate–low-risk PE. All consecutive patients meeting the inclusion criteria during the study period were enrolled.
A total of 129 patients with IHR or HR acute PE were included in the study , of whom 62 were managed with PERT involvement and 67 received standard care without PERT participation.
The study protocol was approved by the Institutional Ethics Committee of the University of Medicine and Pharmacy at Ho Chi Minh City (approval number: 2657/ĐHYD-HĐĐĐ). All patients were provided with written informed consent forms and agreed to participate prior to inclusion and sample collection for the study. Study was performed in compliance with Helsinki 2024 agreement on human studies.
Baseline variables
Baseline demographic and clinical variables collected at the time of PE diagnosis included age, sex, comorbidities (chronic pulmonary disease, heart failure, and active cancer); laboratory findings (D-dimer, troponin, and NT-proBNP) and imaging findings (CTPA and transthoracic echocardiography).
Clinical severity was assessed using the PE Severity Index (PESI) and the 2019 ESC risk stratification for acute PE.
The PESI score is a validated prognostic tool based on demographic characteristics, comorbidities, and clinical findings, used to estimate 30-day mortality risk in patients with acute PE. ESC 2019 risk stratification classifies acute PE according to hemodynamic status, RV dysfunction, and cardiac biomarker elevation (1).
CTPA and transthoracic echocardiography
Imaging was used to assess bilateral PE, thrombotic involvement of the main pulmonary artery (PA) or main left/right PAs, contrast reflux into the inferior vena cava (IVC), RV dilation, RV dysfunction, and the presence of proximal deep vein thrombosis. RV dysfunction was assessed using both CTPA and transthoracic echocardiography according to the 2019 ESC guideline criteria, including an RV/LV diameter ratio >1.0 on CTPA and guideline-defined echocardiographic markers of RV dysfunction.
PERT description
PERT activation was considered for patients with IHR or HR PE; however, final activation depended on clinical characteristics, disease severity, available institutional resources, and the treating physician’s judgment. At our institution, PERT operates continuously (24/7) as a rapid-response, multidisciplinary team. There are two activation pathways depending on patient risk stratification. For IHR patients without hemodynamic instability, an abbreviated PERT model is activated. This core team includes specialists from cardiology, interventional cardiology, and diagnostic imaging. For HR patients with hemodynamic compromise, a full PERT team is engaged, adding cardiothoracic surgery and intensive care specialists. Additional disciplines (e.g., pulmonology, hematology….) may be involved depending on the patient’s clinical condition. PERT can be activated by the attending physician via an internal hotline. The on-call physicians from the relevant departments respond promptly, and opinion of experts can be obtained via phone consultation if necessary. As a rapid-response unit, the PERT typically assembles within 20 minutes of activation and aims to reach a definitive management decision within 30 minutes. Members of the team actively coordinate to facilitate early access to advanced interventions when indicated. PERT determines the most appropriate therapeutic strategy based on each patient's clinical profile. Available treatment options include anticoagulation alone, CDTh, CDT, extracorporeal membrane oxygenation (ECMO), full-dose ST, low-dose ST, and surgical embolectomy. Beyond initial evaluation, the PERT remains involved in inpatient and post-discharge follow-up. Reactivation of the team is possible in cases of clinical deterioration during hospitalization.
Outcome variables and follow-up
PERT-related data, including the treatment decisions and participating specialties for each activation, were also recorded. In-hospital outcomes were collected from the medical record. Advanced therapy was defined as the implement of any CDTh, CDT, full or half dose ST, surgical embolectomy, ECMO. The 30-day post-discharge outcomes were followed using the hospital’s inpatient and outpatient electronic medical record system. For patients who did not return for follow-up at our institution, telephone interviews were conducted to collect clinical outcomes. The composite adverse outcome was defined as all-cause in-hospital mortality, all-cause mortality within 30 days after discharge, or all-cause readmission within 30 days after discharge. Bleeding events were defined as bleeding episodes that required a change in treatment, including temporary or permanent discontinuation of anticoagulation, additional medical intervention, or blood transfusion.
Statistical analysis
Analyses were performed using R. Baseline characteristics are summarized using descriptive statistics. Continuous variables are reported as mean (standard deviation) or median (interquartile range), and categorical variables as frequencies and percentages. Group comparisons were performed using the Chi-square or Fisher’s exact test for categorical variables and the Student’s t-test or Mann–Whitney U test for continuous variables, as appropriate. The association between PERT involvement and the composite adverse outcome was evaluated using log-binomial regression adjusting for pre-specified confounders, with results reported as risk ratios and 95% confidence intervals. Confounding variables were selected a priori based on clinical relevance and prior literature, with additional consideration of variables associated with the outcome in univariable analyses (p<0.05). A two-sided p value <0.05 was considered statistically significant.
Results
A total of 129 patients with IHR or HR acute PE were included in the analysis, of whom 62 (48.1%) were managed with PERT involvement and 67 (51.9%) received standard care without PERT participation.
Table 1 summarizes the baseline demographic and clinical characteristics of the study population. The median age was 66.0 years, and 25.5% of patients were male. Most patients presented with high disease severity, including a high prevalence of RV impairment and occlusion of the main PAs. The majority of patients were classified as IHR according to ESC 2019 criteria.
|
Table 1. Baseline demographic and clinical characteristics of the study population |
|
|
Variables |
Overall (N = 129) |
|
Age, years |
66.0 (53.0 – 75.0) |
|
Male, n(%) |
96 (74.4) |
|
Chronic pulmonary disease, n(%) |
5 (3.9) |
|
Heart failure, n(%) |
33 (25.6) |
|
Cancer, n(%) |
21 (16.3) |
|
D-dimer, ng/ml |
8302.5 (3685.3–20040.8) |
|
Troponin, pg/ml |
75.2 (34.4–164.25) |
|
NT-proBNP, pg/mL |
1995 (865–5857) |
|
Bilateral pulmonary embolism, n(%) |
112 (86.8) |
|
Main pulmonary artery or main left/right pulmonary artery, n(%) |
97 (75.2) |
|
Contrast reflux into IVC, n(%) |
69 (53.5) |
|
Right ventricular dilation, n(%) |
68 (52.7) |
|
Right ventricular dysfunction, n(%) |
43 (33.3) |
|
Proximal DVT, n(%) |
67 (51.9) |
|
ESC 2019 risk of early death |
|
|
Intermediate – high risk, n(%) |
101 (78.3) |
|
High risk, n(%) |
28 (21.7) |
|
PESI, points |
110 (83–134) |
|
PERT involvement, n(%) |
62 (48.1) |
|
Cardiologist, n(%) (N=62) |
61 (98.4) |
|
Interventionist, n(%) (N=62) |
62 (100) |
|
Cardiac surgeon, n(%) (N=62) |
33 (53.2) |
|
Radiologist, n(%) (N=62) |
58 (93.5) |
|
Intensivist, n(%) (N=62) |
31 (50) |
|
Other specialties, n(%) (N=62) |
47 (75.8) |
|
Advanced therapies, n(%) |
45 (34.9) |
|
Full-dose thrombolysis, n(%) |
8 (6.2) |
|
Half-dose thrombolysis, n(%) |
23 (17.8) |
|
Catheter-directed thrombectomy, n(%) |
6 (4.7) |
|
IVC filter, n(%) |
5 (3.9) |
|
Surgical embolectomy, n(%) |
2 (1.6) |
|
Catheter-directed thrombolysis, n(%) |
7 (5.4) |
|
No anticoagulation at discharge, n(%) |
30 (23.3) |
|
In-hospital mortality, n(%) |
13 (10.1) |
|
In-hospital bleeding, n(%) |
38 (29.5) |
|
1-month post-discharge mortality, n(%) |
15 (12.9) |
|
1-month readmission, n(%) |
22 (17.1) |
|
Composite adverse outcome, n(%) |
47 (36.4) |
|
Data are presented as median (IQR) and number (%) DVT - deep vein thrombosis. IVC - inferior vena cava, NT-proBNP – N-terminal pronatriuretic peptide, PERT - pulmonary embolism response team, PESI – pulmonary embolism severity index |
|
As shown in Table 2, the PERT group included a higher proportion of male patients, whereas cancer was more prevalent in the non-PERT group (p=0.018 for both). Overall, patients in the PERT group presented with a more severe clinical profile. Cardiac biomarkers were significantly higher in the PERT group, including troponin and NT-proBNP (p<0.001 and p=0.021, respectively). Radiological and echocardiographic markers of right heart strain were also more frequent, including contrast reflux into the IVC (p<0.001), RV dilation (p<0.001), and RV dysfunction (p=0.036). The proportion of thrombotic occlusion involving the main PA or the main left/right PAs was higher in the PERT group. The proportion of HR PE tended to be higher in the PERT group, while IHR PE was more common in the non-PERT group, although this difference did not reach statistical significance. Advanced therapies were used significantly more often in the PERT group (p < 0.001), particularly ST. In-hospital bleeding was more frequent in the PERT group (p=0.009).
|
Table 2. Comparison of baseline characteristics, interventions, and outcomes between PERT group and non-PERT group |
|||
|
VariableS |
Non-PERT group (N=67) |
PERT group (N=62) |
p |
|
Age, years |
66.5 (55.0–77.0) |
64.0 (51.0–74.0) |
0.222 |
|
Male, n(%) |
44 (65.7) |
52 (83.9) |
0.018 |
|
Chronic pulmonary disease, n(%) |
4 (6.0) |
1 (1.6) |
0.367* |
|
Cancer, n(%) |
23 (34.3) |
10 (16.1) |
0.018 |
|
Heart failure, n(%) |
4 (6.0) |
6 (9.7) |
0.519* |
|
D-dimer, ng/mL |
6282.0 (2793.0–14985.0) |
12586.0 (4518.0–33695.0) |
0.059 |
|
Troponin, pg/mL |
46.2 (27.4–107.0) |
115.5 (54.7–284.0) |
< 0.001 |
|
NT-proBNP, pg/mL |
1520.0 (603.5–4200.5) |
2901.0 (1036.0–7115.0) |
0.021 |
|
Bilateral pulmonary embolism, n(%) |
57 (85.1) |
55 (88.7) |
0.542 |
|
Main pulmonary artery or main left/right pulmonary artery, n(%) |
40 (59.7) |
57 (91.9) |
< 0.001 |
|
Contrast reflux into IVC, n(%) |
25 (37.3) |
44 (71.0) |
< 0.001 |
|
Right ventricular dilation, n(%) |
25 (37.3) |
43 (69.4) |
< 0.001 |
|
Right ventricular dysfunction, n(%) |
17 (26.2) |
26 (44.1) |
0.036 |
|
Proximal DVT, n(%) |
35 (58.3) |
32 (52.5) |
0.516 |
|
High risk, n(%) |
10 (14.9) |
18 (29.0) |
|
|
Intermediate – high risk, n(%) |
57 (85.1) |
44 (71.0) |
0.052 |
|
PESI, points |
105.0 (83.0–131.0) |
112.5 (85.0–139.0) |
0.596 |
|
Advanced therapy, n(%) |
10 (14.9) |
35 (56.5) |
< 0.001 |
|
Systemic thrombolysis, n(%) |
5 (7.5) |
26 (41.9) |
< 0.001 |
|
Half-dose systemic thrombolysis, n(%) |
3 (4.5) |
20 (32.3) |
< 0.001* |
|
Catheter-directed thrombectomy, n(%) |
1 (1.5) |
5 (8.1) |
0.105* |
|
IVC filter, n(%) |
3 (4.5) |
2 (3.2) |
1.000* |
|
Surgical embolectomy, n(%) |
0 (0.0) |
2 (3.2) |
0.229* |
|
Catheter-directed thrombolysis, n(%) |
4 (6.0) |
3 (4.8) |
1.000* |
|
No anticoagulation at discharge, n(%) |
17 (25.4) |
13 (21.0) |
0.554 |
|
In-hospital bleeding, n(%) |
13 (19.4) |
25 (40.3) |
0.009 |
|
In-hospital mortality, n(%) |
7 (10.4) |
6 (9.7) |
0.885 |
|
1-month post-discharge mortality, n(%) |
10 (14.9) |
5 (8.1) |
0.347 |
|
1-month readmission, n(%) |
16 (23.9) |
6 (9.7) |
0.032 |
|
Composite adverse outcome, n(%) |
31 (46.3) |
16 (25.8) |
0.016 |
|
Data are presented as median (IQR) and number (%) *Fischer exact test, Chi-square test and Mann Whitney test DVT - deep vein thrombosis. IVC - inferior vena cava, NT-proBNP – N-terminal pronatriuretic peptide, PERT - pulmonary embolism response team, PESI – pulmonary embolism severity index |
|||
As shown in table 3, no adverse composite outcome group demonstrated a higher utilization of PERT (p=0.026) and advanced therapeutic strategies compared with those in adverse composite outcome group (41.5% vs. 23.4%); however, this difference did not reach statistical significance (p=0.06). CDT was used significantly more frequently in the better outcome group (8.5% vs. 0.0%, p = 0.047). There was no significant difference in bleeding rates between the two groups.
|
Table 3. Comparison of baseline characteristics and interventions between no adverse composite outcome and adverse composite outcome groups |
|||
|
Variable |
No adverse composite outcome (N=82) |
Adverse composite outcome (N=47) |
p |
|
Age, |
69.0 (63.0–80.0) |
61.0 (51.0–72.0) |
0.003 |
|
Male, n(%) |
18 (22.0) |
15 (31.9) |
0.299 |
|
Chronic pulmonary disease, n(%) |
3 (3.7) |
2 (4.3) |
1.000 |
|
Heart failure, n(%) |
5 (6.1) |
5 (11.1) |
0.600 |
|
Cancer, n(%) |
13 (15.9) |
8 (17.0) |
1.000 |
|
Bilateral pulmonary embolism, n(%) |
75 (91.5) |
37 (78.7) |
0.074 |
|
Main pulmonary artery or main left/right pulmonary artery, n(%) |
65(79.3) |
32 (68.1) |
0.228 |
|
Contrast reflux into IVC, n(%) |
49 (59.8) |
20 (42.6) |
0.089 |
|
Right ventricular dilation, n(%) |
49 (59.8) |
19 (40.4) |
0.053 |
|
Right ventricular dysfunction, n(%) |
31 (37.8) |
12 (28.6) |
0.410 |
|
Proximal DVT, n(%) |
45 (56.2) |
22 (53.7) |
0.938 |
|
High risk, n(%) |
10 (14.9) |
18 (29.0) |
|
|
Intermediate – high risk, n(%) |
57 (85.1) |
44 (71.0) |
0.052 |
|
PESI, points |
105.0 (83.0–131.0) |
112.5 (85.0–139.0) |
0.596 |
|
NT-proBNP, pg/ml |
2589.0 (885.0–6181.0) |
1737.0 (818.8–4820.8) |
0.557 |
|
Troponin, pg/ml |
81.3 (29.0–155.1) |
72.7 (37.4–172.3) |
0.711 |
|
D-dimer, ng/ml |
7750.0 (4340.0–18946.0) |
8855.0 (2972.0–28140.0) |
0.797 |
|
PERT involvement, n(%) |
46 (56.1) |
16 (34.0) |
0.026 |
|
Advanced therapies, n(%) |
34 (41.5) |
11 (23.4) |
0.060 |
|
Systemic thrombolysis, n(%) |
22 (26.8) |
9 (19.1) |
0.442 |
|
Half-dose systemic thrombolysis, n(%) |
17 (20.7) |
6 (12.8) |
0.369 |
|
Catheter-directed thrombectomy, n(%) |
4 (4.9) |
2 (4.3) |
1.000* |
|
IVC filter, n(%) |
4 (4.9) |
1 (2.1) |
0.652* |
|
Surgical embolectomy, n(%) |
2 (2.4) |
0 (0.0) |
0.533* |
|
Catheter-directed thrombolysis, n(%) |
7 (8.5) |
0 (0) |
0.047* |
|
No anticoagulation at discharge, n(%) |
7 (8.5) |
23 (48.9) |
< 0.001 |
|
In-hospital bleeding, n(%) |
27 (32.9) |
11 (23.4) |
0.347 |
|
Data are presented as median (IQR) and number (%) *Fischer exact test, Chi-square test and Mann Whitney test DVT - deep vein thrombosis. IVC - inferior vena cava, NT-proBNP – N-terminal pronatriuretic peptide, PERT - pulmonary embolism response team, PESI – pulmonary embolism severity index |
|||
In the adjusted analysis, PERT involvement was independently associated with a lower risk of the composite adverse outcome (RR 0.58, 95% CI 0.38–0.90), whereas higher PESI score was associated with an increased risk. Absence of anticoagulation at discharge emerged as a strong independent predictor of adverse outcomes (RR 2.73, 95% CI 1.76–4.23) (Fig. 1).
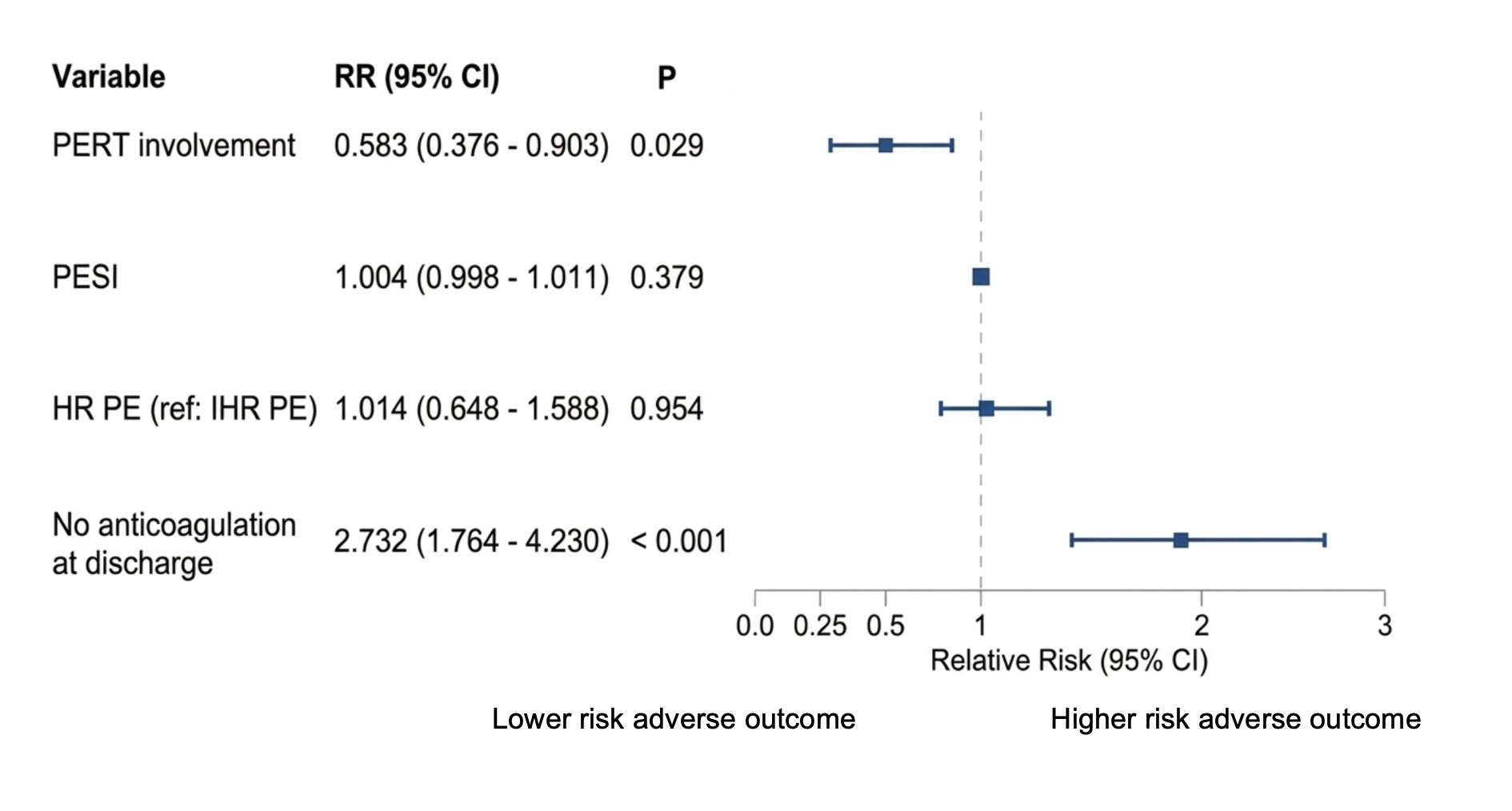
Figure 1. Forest plot of factors associated with the composite adverse outcome (multivariable log-binomial regression)
CI – confidence interval, HR- High risk, IHR- Intermediate-high risk, PE- Pulmonary embolism, PERT- Pulmonary Embolism Response Team, RR – relative risk
Discussion
Our observational single-center study shows that involvement of PERT in the management of acute PE is associated with a 42% relative reduction in the risk of early adverse outcomes (RR 0.58, 95% CI 0.38–0.90). Patients managed with PERT had a lower risk of the composite endpoint, comprising in-hospital mortality, 30-day post-discharge mortality, or 30-day readmission, compared with those managed without PERT involvement. The observed reduction in the composite adverse outcome indicates that PERT delivers a broader benefit, from survival to readmission in critical risk classification. In our research, a 36.4% composite event rate means that over one-third of these patients either died or required rehospitalization shortly after PE diagnosis. This reflects the severity of IHR and HR PE and the challenges in management.
Our observation is partly in line with outcomes reported in similar PE cohorts. In prior literature, Lacey et al. (8) conducted a prospective study at a Cleveland center (USA) involving 220 patients with all risk PE, and were among the first to apply a long-term 90-day composite endpoint that included mortality, readmission, and major bleeding. The study found that activation of a PERT—initiated in 21% of cases—was associated with a 60% reduction in the risk of this 90-day composite outcome compared to non-activation (HR = 0.40; p < 0.01) (8). However, not all reports have shown a definitive mortality benefit. In contrast, Annabathula et al. (9) found no significant difference in 30-day readmission rates before and after PERT implementation (15.0% vs. 15.8%). However, their analysis included an unselected PE population, rather than focusing specifically on IHR and HR patients—where the impact of PERT is likely to be more pronounced (9). Moreover, benefits of PERT are multifaceted and may extend across several domains, including symptom improvement, reduction in bleeding, and mortality. As a result, studies that focus on a single outcome may fail to capture the overall clinical impact of a multidisciplinary PERT approach. This may partly explain the discrepant findings across previous studies.
A possible explanation for this benefit in composite adverse outcome is that patients managed with PERT were substantially more likely to receive advanced therapeutic interventions especially ST compared with those managed without PERT involvement (Table 2). This finding is consistent with prior reports from international PERT studies, which have shown that PERT implementation is associated with increased utilization of advanced therapies in appropriately selected patients (5, 6, 10). These interventions have been consistently shown to achieve more rapid and pronounced reductions in thrombotic burden and pulmonary vascular obstruction compared with anticoagulation alone (11, 12). Importantly, the clinical benefits of thrombus burden reduction extend beyond mortality reduction; they are closely linked to improvements in RV recovery, exercise capacity, symptom burden (e.g., dyspnea and fatigue), and health-related quality of life, as well as a potential reduction in post–PE syndrome. Consequently, evaluating PERT effectiveness solely on the basis of mortality may fail to capture these clinically meaningful benefits. A composite outcome may therefore provide a more sensitive and comprehensive assessment of the true clinical impact of PERT-based management. The favorable outcomes associated with PERT in our study may reflect the high level of multidisciplinary engagement at our institution, with consistent participation from key specialties such as cardiology (98.4%), radiology (93.5%), interventional cardiology (100%), and cardiothoracic surgery (53.2%). These factors could help explain partly why some prior studies did not demonstrate similar benefits, particularly those with less integrated team structures.
Despite greater disease severity in the PERT group (higher cardiac biomarker levels, more frequent RV dysfunction, and a higher prevalence of thrombotic involvement of the main PA or main left/right PA), PERT involvement was associated with a lower risk of the composite adverse outcome in our study, supporting the appropriateness of patient selection at our institution. Selective activation in patients with the above factors allowed PERT to deliver meaningful clinical benefit while conserving institutional resources. In contrast, more routine activation would dilute its clinical impact and lead to inefficient resource utilization. Consistently, other studies report that PERT activations tend to be triggered for more severe presentations, such as central (“saddle” or main PA) emboli or RV dysfunction (10). Centers that included a broader mix of lower-risk PE might observe smaller mortality and morbidity differences. Patients with active cancer were more often managed without PERT involvement, likely reflecting real-world clinical decision-making due to advanced disease, limited expected benefit from aggressive treatment, or higher bleeding risk, rather than a predefined exclusion of the study.
The bleeding rate was significantly higher in the PERT group, this signal is explained by the greater use of advanced reperfusion strategies—particularly ST—among PERT-managed patients (Table 2). However, as shown in Table 3, bleeding rates were comparable between patients who did and did not experience the composite adverse outcome, suggesting that the excess bleeding observed in the PERT arm did not translate into a higher incidence of the adverse composite endpoint. This finding showed that, while bleeding is more frequently observed when advanced reperfusion strategies are used, such events may be clinically manageable and may not offset the overall benefits when decisions are guided by an experienced PERT. Overall, published PERT studies have generally reported no significant change in bleeding rates, even though PERT implementation or activation is often associated with greater use of advanced therapies (6, 13, 14). In our study, in-hospital bleeding was higher in the PERT group, which may be explained by a more aggressive reperfusion strategy, as 29.4% of patients received fibrinolysis-related therapies and reflect the variation in PERTs practice.
The 2026 AHA/ACC guideline (15) recommends PERT for patients with acute PE at increased risk of adverse outcomes (Class I, Level B). Importantly, the guideline also emphasizes that the composition and activation of PERTs are not standardized and should be adapted to local institutional resources, which may partly explain the heterogeneous findings across prior studies. Our study provides additional insight into this evidence gap by describing the real-world operational structure of a PERT, the clinical features associated with PERT activation, and the management decisions. These findings may be useful for centers seeking to develop PERT in the future.
We chose a composite adverse outcome because our sample was very limited (only 129 cases over 5 years). With such small numbers, analyzing each individual outcome such as mortality, or readmission separately would lack statistical power, risking false-negative results. Using a composite endpoint increases the event rate to detect any overall benefit of the PERT intervention. This sample size is appropriate for a single-center study focusing exclusively on the critical risk classifications. For comparison, the PERT CELZAT registry in Poland reported 110 HR and IHR patients over 5 years (16).
Similarly, when specifically isolating the HR and IHR cohorts in the National PERT Consortium Registry (USA), the annual volume per center typically fluctuates between 10 and 20 cases (17).
In multivariate regression model, PESI score included multiple clinical factors. Therefore, its inclusion obviated the need to adjust separately for each clinical parameter. ESC risk category was included to account for imaging and biomarker-based risk stratification. PESI and ESC risk category captured different dimensions of risk. Therefore, both variables were retained a priori in the multivariable model. Anticoagulation at discharge was incorporated given its strong biological plausibility with early adverse outcomes. Although CDT was significantly associated with fewer adverse outcomes in univariable analysis, no CDT procedure was applied among patients with adverse outcome (Table 3). Therefore, CDT could not be retained in the multivariable model. This finding should be interpreted with caution and does not indicate a definitive protective effect of CDT; rather, it reflects the limited sample size.
Study limitations
This study has several limitations. First, as a single-center investigation with a relatively modest sample size, our findings may not be generalizable to all hospital settings. Given the non-randomized observational design, our study remains susceptible to selection bias. Although we attempted to mitigate confounding variables through multivariable regression adjustment, residual confounding and selection bias may still be present.
Conclusions
In this observational single-center, PERT involvement was associated with a lower risk of the composite adverse outcome — defined as all-cause in-hospital mortality, or all-cause mortality or readmission within 30 days after discharge — despite greater baseline disease severity in the PERT group among patients with IHR and HR PE. This benefit suggests that PERT influences the patient care from hospitalization to the early post-discharge period. Our findings support a selective activation strategy, focusing PERT on patients with significant elevated cardiac biomarkers and clear evidence of RV strain on echocardiography and CTPA, and thrombotic occlusion involving the main PA, which may maximize clinical impact while conserving institutional resources and reducing unnecessary workload. Although bleeding events were more frequent in the PERT group, this did not translate into higher mortality.
Ethics: The study protocol was approved by the Institutional Ethics Committee of the University of Medicine and Pharmacy at Ho Chi Minh City (approval number: 2657/ĐHYD-HĐĐĐ). All patients were provided with written informed consent forms and agreed to participate prior to inclusion and sample collection for the study. Study was performed in compliance with Helsinki 2024 agreement on human studies.
Peer-review: External and Internal
Conflict of interest: None to declare
Authorship: The authors acknowledge the contributions of all team members to this study: conceptualization by DT Bui; data curation by HT Nguyen, MNT Nguyen, HT Nguyen, and HV Nguyen; formal analysis by KM Le and VH Vu; methodology by H Tran, DH Nguyen, and TT Au; software by KD Tran; investigation by HT Nguyen; writing of the original draft by KD Tran; and writing, review, and editing by DT Bui, VH Vu, HV Nguyen, H Tran, MTT Nguyen, DG Nguyen, TT Au, KM Le, HT Nguyen, HT Nguyen, and KD Tran. Thus equally contributed to the study and manuscript preparation and fulfilled all authorship criteria.
Acknowledgements and Funding: This research was financially supported by the Ho Chi Minh City Science and Technology Development Fund under grant number 104/2024/HĐ-QKHCN.
Statement on A.I.-assisted technologies use: Artificial intelligence tools were used only for language editing. All scientific content, data analysis, and interpretation were performed by the authors, who take full responsibility for the integrity and accuracy of the work.
Data and material availability: The de-identified individual participant data that support the findings of this study are available from the corresponding author upon reasonable request. Any share should be in frame of fair use, with acknowledgement of source and/ or collaboration
References
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

