Atrial septal defect patients with greater shunts show susceptibility for ventricular arrhythmias
ORIGINAL RESEARCH ARTICLE
Atrial septal defect patients with greater shunts show susceptibility for ventricular arrhythmias
Article Summary
- DOI: 10.24969/hvt.2020.206
- Page(s): 92-98
- CARDIOVASCULAR DISEASES
- Published: 07/07/2020
- Received: 13/06/2020
- Revised: 07/07/2020
- Accepted: 07/07/2020
- Views: 10910
- Downloads: 7770
-
Citations
- Keywords: atrial septal defect, electrocardiogram, ventricular arrhythmia, risk, ventricular repolarization
PDF PRINT VERSION CommentsAddress for CorrespondenceAddress for Correspondence: Osman Can Yontar, University of Health Sciences Turkey, Samsun Training and Research Hospital, Cardiology Clinic, Samsun, Turkey Email: drcanyontar@gmail.com
Osman Can YontarUniversity of Health Sciences Turkey, Samsun Training and Research Hospital, Cardiology Clinic, Samsun, Turkey
Abstract
Objective: Ventricular arrhythmia episodes are not infrequent in patients with atrial septal defect (ASD). Disturbance in cardiac volume and pressures may lead to enlargement and fibrosis in heart. An interatrial volume displacement through septal defect, briefly interatrial shunt, is the major reason for this complication. Prolongation of the interval between the peak and end of the T wave (Tpeak to Tend, Tp-e) on the 12-lead electrocardiogram (ECG), is utilized as a marker of ventricular arrhythmogenesis during last years. The aim of this study was to assess if there is an impact of shunt ratio on ventricular repolarization in patients with ASD by using Tp-e interval, Tp-e/QT ratio, and Tp-e/QTc ratio.
Methods: Patient records of Samsun Training and Research Hospital were retrospectively analyzed. Electrocardiograms of 133 patients, who were diagnosed as ASD between January 2016 and December 2019 were obtained and scanned. ECG intervals were measured. Shunt ratios, right ventricle diameters and volumes were also acquired. Patients were grouped into two by their calculated shunt ratio, ratio of ≥2.0 is accepted as a high shunt group and <2.0 as a low shunt group.
Results: Both groups’ baseline characteristics were similar. Right ventricular dimensions and systolic pulmonary artery pressure were higher in high shunt group. Furthermore, ASD patients with higher shunt ratio had significantly higher ECG measurements than controls, Tp-e: 103.0 (22.1) vs 76.2 (10.2); Tp-e/QT: 0.25 (0.03) vs 0.21 (0.02); Tp-e/QTc: 0.22 (0.03) vs, 0.17 (0.02); for all p<0.001). Of all ECG parameters; Tp-e (r=0.631, p<0.001), Tp-e/QT (r=0.531, p<0.001) and Tp-e/QTc (r=0.614, p<0.001) had moderate correlation with shunt ratio.
Conclusion: T wave peak-to-end interval is a measure of transmural dispersion of repolarization and accepted as a surrogate for increased ventricular arrhythmogenesis risk. Our findings show that ASD patients whose shunt ratio are ≥2.0 show increased risk for arrhythmias.
Key words: atrial septal defect, electrocardiogram, ventricular arrhythmia, risk, ventricular repolarization
Introduction
Atrial septal defect (ASD) constitutes a significant amount of congenital heart diseases (1). Secundum ASD is the most common type of ASD that causes diversion of blood from systemic to pulmonary circulation. In early stages of life, most patients are asymptomatic. Although some patients are diagnosed early, many of them remain silent until adulthood (2). If the disease stays unnoticed, decrease in exercise capacity, miscellaneous supraventricular arrhythmias, right heart failure, and pulmonary arterial hypertension may occur within years (3, 4). ASD may lead to disruptions in cardiac kinetics dependent on the defect diameter and shunt volume. Shunts which are calculated to be over 2.0 indicate large ASDs with significant blood diversion from left to right atria (5). If atrial and ventricular myocardium is exposed to volume overload, shear stress extends to not only myofibers but also conduction system of heart. Disruptions may manifest as conduction delay within atrium and ventricles (6, 7). Furthermore, the degeneration of cardiac microarchitecture, including enhanced fibrosis, improperly modifies myocardial repolarization (6, 7).
T wave peak to end (Tp-e) interval is recent marker for ventricular susceptibility for arrhythmias and repolarization heterogeneity (8-10). A prolonged Tp-e interval represents a period of susceptibility to ventricular arrhythmias. It was shown that this interval has been associated with increased risk of mortality in various congenital and structural heart diseases (11-15).
There is little information in current literature concerning ventricular arrhythmia susceptibility in ASD.
Our goal is to assess if there is an impact of shunt ratio on transmural repolarization in patients with ASD by using Tp-e interval, Tp-e/QT ratio, and Tp-e/QTc ratio.
Methods
Study participants
Virtual data management system of Samsun Training and Research Hospital was retrospectively analyzed. One hundred and thirty- three patients who had been diagnosed with secundum ASD at outpatient clinic were enrolled. Their electrocardiograms were acquired and scanned. Echocardiographic calculations such as shunt ratio, pulmonary artery pressure and other measurements of right ventricle diameters were gained from previous reports in digital archive. Patients with significant coronary artery lesion, critically ill ASD patients (requiring pulmonary arterial hypertension-specific therapy), moderate or more severe degree valve disease other than tricuspid valve, left ventricular failure, any ASD type other than secundum ASD, left ventricular hypertrophy, atrial fibrillation, right or left bundle block or patients with implanted pacemaker or cardioverter/defibrillator were excluded. Informed consent was obtained from all patients before procedures. Study protocol was endorsed by local scientific board.
Availability of Data and Materials
Supplementary material consisting of supportive data is available from the corresponding author, (OCY), upon reasonable request.
Measurement of Tp-e, QT and QRS Intervals from the 12-Lead ECG
All ECGs were scanned. Utilization of a manual measuring tool and a magnifier for interval measurements; could be either inaccurate or time consuming. ECG papers were scanned and that allowed researcher to make measurements in virtual environment. All intervals were measured by blinded colleague with a virtual caliper on computer interface. Measurements of Tp-e interval were performed from precordial lead V5 as it was described (Fig. 1) (16). From the peak (or the nadir if it was negative) of T wave to the end point where it reaches isoelectric line was measured for Tp-e interval.
The QT interval was measured from the beginning of the QRS complex to where T waves descend onto the isoelectric baseline. When an U wave interrupted the T wave before returning to isoelectric line, the QT interval was measured to the nadir of the bent segment between the T and U waves. The QTc interval was calculated using the Bazett formula: QTc (ms)=QT measured/√RR (sec). QT interval was measured from precordial V5 lead.
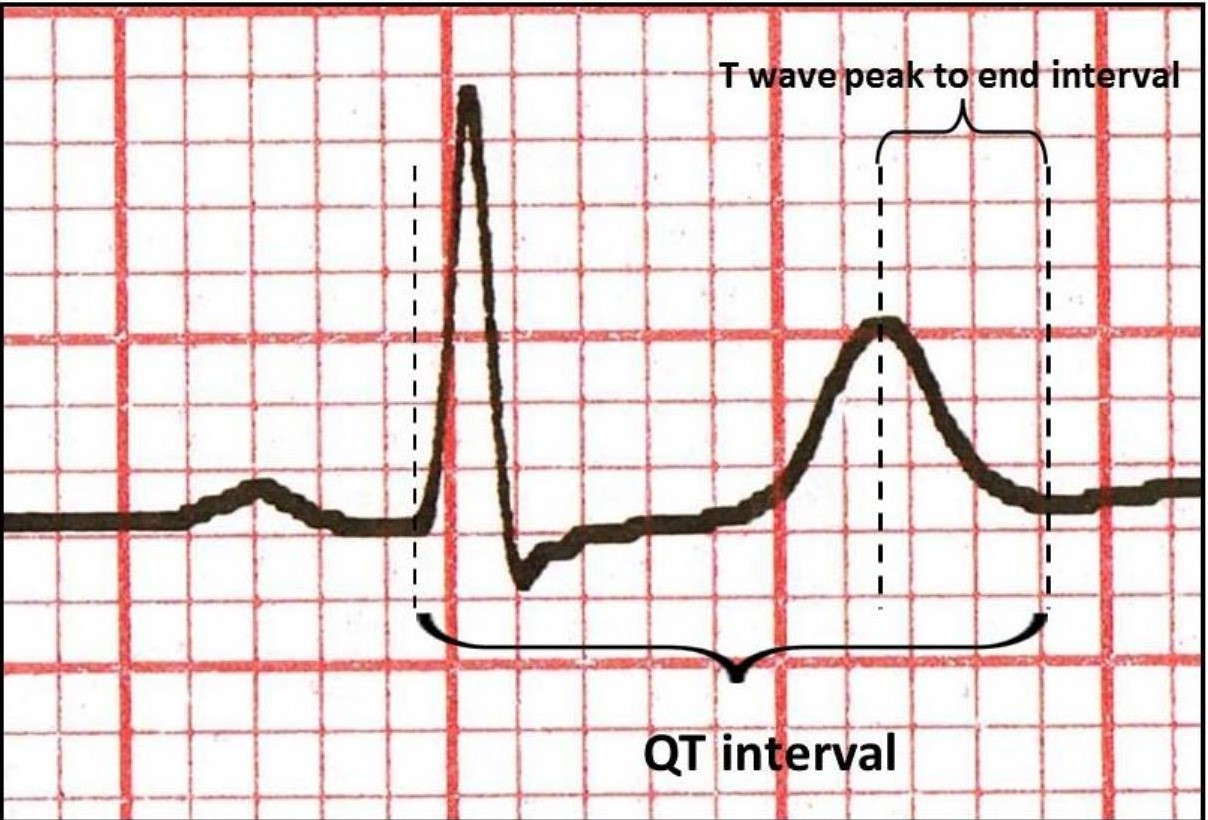
Figure 1. Measurement of intervals on electrocardiogram
Echocardiography
Atrial septal defect shunt ratio was calculated noninvasively by echocardiography (17). Pulmonary perfusion (Qp) was calculated by multiplying right ventricle outflow tract (RVOT) time-velocity integral (in centimeters) and pi number and square of the half of the RVOT diameter (in millimeters). Systemic perfusion (Qs) was calculated by multiplying left ventricular outflow tract time-velocity integral (in centimeters) and pi number and square of the half of the left ventricular outflow tract diameter (in millimeters). Then Qp was divided by Qs to calculate shunt ratio.
Also right ventricular midline/baseline/ end-diastolic base-to-apex distance, end-diastolic and end-systolic volumes, systolic pulmonary artery pressure (SPAP), left ventricular ejection fraction and tricuspid annular plane systolic excursion (TAPSE) were determined. Each echocardiogram was evaluated by 2 blinded sonographers.
Statistical analysis
All analyses were performed using SPSS V 22.0 for Windows (SPSS Inc., Chicago, Illinois, USA). We calculated the minimum number of the individuals that should be sampled with 90% power and 0.05 Type-I error as at least 58 (R 3.0.1. open source program). For parametric and continuous variables, independent-samples t test; for nonparametric and categorical variables, Chi-square test was used for univariate case-control comparisons for all cases vs. controls, respectively. A P value of <0.05 was considered as significant. Spearman correlation coefficient was used for correlation analysis. Association of different variables with electrocardiographic and echocardiographic parameters and shunt ratio were separately calculated in univariate analysis.
Results
Baseline data
A total of 133 patients were grouped into two: 65 patients constituted low shunt ratio group (<2.0), remaining 68 patients formed the group with high shunt ratio (≥2.0). Mean age for patients with high shunt ratio was 40.5 (12.5) and for lower shunt group was 38.9 (12.7) (p=0.463). High shunt group consisted of 47 female patients (72.3%) whereas there were 44 female patients (64.7%) in low shunt group (p=0.358). Twenty three patients (35.4%) of low shunt group had diabetes whereas there were 14 (20.6%) patients with diabetes in high shunt group (p=0.834). High and low shunt groups did not differ in entity of hypertensive individuals (7 vs. 10) and smokers (31 vs. 32).
ECG and echocardiographic variables
Echocardiographic and ECG measurements are listed in Table 1. High shunt group had significantly longer Tp-e, QT and QTc intervals and higher Tp-e/QT and Tp-e/QTc ratios than low shunt group (p<0.001 for all).
Echocardiographic measurements of RV diameters (end-diastolic annulus line diameter, end-diastolic mid-line diameter, end-diastolic base-to-apex distance), RV volumes and SPAP were higher (p<0.001), while TAPSE was significantly lower (p<0.001) in high shunt group as compared to low shunt group. Mean left ventricular ejection fraction was similar in both high and low shunt groups (p>0.05).
Correlations between parameters of transmural repolarization with ECG and echocardiographic variables
Of all ECG parameters; Tp-e (r=0.631, p<0.001), Tp-e/QT (r=0.531, p<0.001) and Tp-e/QTc (r=0.614, p<0.001) had moderate correlation with shunt ratio (Table 2). On the other hand, right ventricular end-diastolic (r=0.783, p<0.001) and end-systolic volumes (r=0.833, p<0.001), TAPSE (r=-0.638, p<0.001) and SPAP (0.677, p<0.001) showed strong correlation with shunt ratio (Table 3).
Discussion
Our results demonstrated that ASD patients whose shunt ratio are ≥2.0 had longer Tp-e intervals and higher Tp-e/QT and Tp-e/QTc ratios than individuals with a lower shunt ratio and there is a positive correlation between shunt ratio and Tp-e interval, Tp-e/QT and Tp-e/QTc ratios
Atrial septal defect is one of the most common congenital heart diseases. Frequency of atrial tachyarrhythmias and conduction diseases is very well studied in this population (18-20). Atrial arrhythmia prevalence shows correlation with size of shunt and occurrence of hemodynamic complications such as pulmonary artery hypertension, as well as with other comorbidities. However, there is a lack of scientific data regarding occurrence of ventricular arrhythmogenesis in this group of patients. Eryu et al. (21) investigated the RR and QT interval variability in a group of ASD patients and found that there was a correlation between these indices and shunt ratio.
Table 1. Comparison of echocardiographic and electrocardiographic parameters between both groups
Parameters
Shunt ratio ≥2.0
(n=68)
Shunt ratio <2.0
(n=65)
p
Mean shunt ratio
2.2 (0.1)
1.3 (0.2)
<0.001
RV1, mm
35.5 (2.4)
32.4 (1.7)
<0.001
RV2, mm
28.0 (2.2)
24.7 (1.4)
<0.001
RV3, mm
66.5 (2.8)
64.0 (1.9)
<0.001
RVEDV, ml
185.4 (13.9)
147.62 (7.0)
<0.001
RVESV, ml
88.46 (7.5)
51.8 (5.4)
<0.001
SPAP, mmHg
35.5 (7.6)
19.3 (5.2)
<0.001
TAPSE, mm
11.2 (2.9)
16.2 (1.6)
<0.001
LVEF, %
60.9 (3.7)
61.0 (4.0)
0.859
QT, msec
409.9 (62.7)
361.4 (35.7)
<0.001
QTc, msec
459.7 (44.0)
428.2 (43.1)
<0.001
Tp-e, msec
103.0 (22.1)
76.2 (10.2)
<0.001
Tp-e/QT ratio
0.25 (0.03)
0.21 (0.02)
<0.001
Tp-e/QTc ratio
0.22 (0.03)
0.17 (0.02)
<0.001
Data are presented as mean (SD)
LVEF - left ventricular ejection fraction, ml - milliliter, mm - millimeter, msec - millisecond, QTc - corrected QT, RV1 - right ventricular end-diastolic annulus line diameter, RV2 - right ventricular end-diastolic mid-line diameter, RV3 - right ventricular end-diastolic base-to-apex distance, RV EDV - right ventricular end-diastolic volume, RVESV - right ventricular end-systolic volume, SPAP - systolic pulmonary artery pressure, TAPSE - tricuspid annular plane systolic excursion, Tp-e- T wave peak to end interval
Table 2. Correlations between electrocardiographic parameters and shunt ratio
Variables
Shunt ratio
r
p
QT, msec
0.457
<0.001
QTc, msec
0.375
<0.001
Tpe, msec
0.631
<0.001
Tpe/QT
0.531
<0.001
Tpe/QTc
0.614
<0.001
Tp-e - T wave peak to end interval, msec - millisecond, QTc - corrected QT
Table 3. Correlations between echocardiographic parameters and shunt ratio
Variables
Shunt ratio
r
p
RV1
0.553
<0.001
RV2
0.569
<0.001
RV3
0.393
<0.001
RVEDV
0.783
<0.001
RVESV
0.833
<0.001
LVEF
0.008
0.926
SPAP
0.677
<0.001
TAPSE
-0.638
<0.001
LVEF - left ventricular ejection fraction, ml - milliliter, mm - millimeter, msec - millisecond, RV1 - right ventricular end-diastolic annulus line diameter, RV2 - right ventricular end-diastolic mid-line diameter, RV3 - right ventricular end-diastolic base-to-apex distance, RVEDV - right ventricular end- diastolic volume, RVESV - right ventricular end systolic volume, SPAP - systolic pulmonary artery pressure, TAPSE - tricuspid annular plane systolic excursion
Authors came to a conclusion that these repolarization indices provided information on alteration of sinus node autonomic control and the pathophysiology of myocardial repolarization. A more recent study (22) was able to show prolonged QT intervals in children with relevant hemodynamic impairment. Authors reach to a conclusion that after closure operation patients’ QTc interval shortened significantly. This finding may create a discussion point for our article’s intellectual basis. In our study, we utilized another myocardial repolarization index, the Tp-e interval, in concordance with QT interval, to determine ventricular arrhythmia susceptibility in a group of ASD patients with relatively lower shunt ratio.
Miscellaneous methods were utilized in order to assess myocardial repolarization including QT dispersion (QTd), corrected QT dispersion (cQTd), and transmural dispersion of repolarization. Recent studies demonstrated that Tp-e interval can be used as an index of total (transmural, apico-basal, and global) repolarization disparity (23, 24). Increased Tp-e interval might be a useful index to predict ventricular tachyarrhythmias and cardiovascular mortality (25). Furthermore, Tp-e/QT ratio has been suggested to be a more accurate measure for the dispersion of ventricular repolarization compared to QTd, cQTd, and Tp-e intervals, which is independent of alterations in heart rate (26). Also, these markers may be used as an ECG indexes of ventricular arrhythmogenesis and sudden cardiac death (16, 20). Previous studies showed that prolongation of Tp-e interval was associated with increased mortality in various clinical cardiac conditions (11, 12, 14, 16). The novel repolarization indexes Tp-e interval and Tp-e/QT ratio, has not been studied in ASD patients before.
Abnormal hemodynamics in ASD patients may play the most important role in ventricular arrhythmogenesis. Left-to-right shunt increases right atrial and eventually right ventricular volume and pressure load to some extent (1). Abnormal right ventricle workload may promote fibrosis in myocardial tissue that would precede an arrhythmic risk. On the other hand, right ventricular volume and pressure overload shift the interventricular septum towards left ventricle. This displacement precedes an impaired diastolic filling and a reduced stroke volume. This would also provoke fibrosis in ventricle. Our patients with larger shunts had their right ventricular end-systolic and end-diastolic volumes significantly increased, which clearly indicates right heart failure. Another theory for conduction heterogeneity in these patients is that, left ventricular diastolic impairment might lead to autonomic imbalance (27) which would directly affect Tp-e and related intervals. Eryu et al (21) asserted that there was an increase in sympathetic control of the heart or a decrease in parasympathetic control of the heart in children with ASD. In our opinion there should be other factors in charge which involve microarchitecture and ion channels and signal transmission than macro changes in heart chambers.
Study limitations
The most important restriction of our study is the limited number of patients. Another limitation we did not assess the association between ventricular arrhythmias with Tp-e interval and Tp-e/QT ratio. Also study population could not be followed-up prospectively for ventricular arrhythmic episodes. Large-scale prospective studies are needed to determine the predictive value of prolonged Tp-e interval and increased Tp-e/QT ratio in this population.
Conclusion
Our study revealed that transmural repolarization is more deteriorated in ASD patients with large shunts and dilated right ventricle than individuals with a lower shunt ratio. Tp-e interval and Tp-e/QT ratio might be a useful marker of arrhythmia susceptibility in this group.
Peer-review: External and internal
Conflict of interest: None to declare
Authorship: O.C.Y
Acknowledgments and funding: None to declare
References
1. Van der Linde D, Konings EEM, Slager MA, Witsenburg M, Helbing WA, Takkenberg JJ, et al. Birth prevalence of congenital heart disease worldwide: a systematic review and meta-analysis. J Am Coll Cardiol 2011; 58:2241-7. https://doi.org/10.1016/j.jacc.2011.08.025 PMid:22078432 2. Kuijpers JM, Mulder BJ, Bouma BJ. Secundum atrial septal defect in adults: a practical review and recent developments. Neth Heart J 2015; 23:205-11. https://doi.org/10.1007/s12471-015-0663-z PMid:25884091 PMCid:PMC4368528 3. Moons P, Bovijn L, Budts W, Belmans A, Gewillig M. Temporal trends in survival to adulthood among patients born with congenital heart disease from 1970 to 1992 in Belgium. Circulation 2010; 122: 2264-72. https://doi.org/10.1161/CIRCULATIONAHA.110.946343 PMid:21098444 4.Yong G, Khairy P, Guise PD, Dore A, Marcotte F, Mercier LA, et al. Pulmonary arterial hypertension in patients with transcatheter closure of secundum atrial septal defects a longitudinal study. Circ Cardiovasc Interv 2009; 2:455-62. https://doi.org/10.1161/CIRCINTERVENTIONS.108.826560 PMid:20031756 5.Geva T, Martins JD, Wald RM. Atrial septal defects. Lancet 2014; 383: 1921-32. https://doi.org/10.1016/S0140-6736(13)62145-5 6. Chubb H, Whitaker J, Williams SE, Head CE, Chung NA, Wright MJ, et al. Pathophysiology and management of arrhythmias associated with atrial septal defect and patent foramen ovale. Arrhythm Electrophysiol Rev 2014; 3: 168-72. https://doi.org/10.15420/aer.2014.3.3.168 PMid:26835086 PMCid:PMC4711537 7. Morton JB, Sanders P, Vohra JK, Sparks PB, Morgan JG, Spence SJ, et al. Effect of chronic right atrial stretch on atrial electrical remodeling in patients with an atrial septal defect. Circulation 2003; 8: 1775-82. https://doi.org/10.1161/01.CIR.0000058164.68127.F2 PMid:12665497 8. Taggart P, Sutton PM, Opthof T, Coronel R, Trimlett R, Pugsley W, et al. Transmural repolarization in the left ventricle in humans during normoxia and ischaemia. Cardiovasc Res 2001; 50: 454 - 62. https://doi.org/10.1016/S0008-6363(01)00223-1 9. Opthof T, Coronel R, Janse MJ. Is there a significant transmural gradient in repolarization time in the intact heart?: Repolarization gradients in the intact heart. Circ Arrhythm Electrophysiol. 2009; 2: 89-96. https://doi.org/10.1161/CIRCEP.108.825356 PMid:19808447 10. Antzelevitch C, Sicouri S, Litovsky SH, Lukas A, Krishnan SC, Di Diego JM, et al. Heterogeneity within the ventricular wall. Electrophysiology and pharmacology of epicardial, endocardial, and M cells. Circ Res 1991; 69: 1427-9. https://doi.org/10.1161/01.RES.69.6.1427 PMid:1659499 11.Topilski I, Rogowski O, Rosso R, Justo D, Copperman Y, Glikson M et al. The morphology of the QT interval predicts torsade de pointes during acquired bradyarrhythmias. J Am Coll Cardiol 2007; 49: 320-8. https://doi.org/10.1016/j.jacc.2006.08.058 PMid:17239713 12. Shimizu M, Ino H, Okeie K, Yamaguchi M, Nagata M, Hayashi K, et al. T-peak to T-end interval may be a better predictor of high-risk patients with hypertrophic cardiomyopathy associated with a cardiac troponin I mutation than QT dispersion. Clin Cardiol 2002; 25: 335-9. https://doi.org/10.1002/clc.4950250706 PMid:12109867 PMCid:PMC6653869 13. Yontar OC, Karaagac K, Tenekecioglu E, Tutuncu A, Demir M, Melek M. Assessment of ventricular repolarization inhomogeneity in patients with mitral valve prolapse: value of T wave peak to end interval. Int J Clin Exp Med 2014; 7: 2173-8. 14. Haarmark C, Hansen PR, Vedel-Larsen E, Pedersen SH, Graff C, Andersen MP, et al. The prognostic value of the Tpeak-Tend interval in patients undergoing primary percutaneous coronary intervention for ST-segment elevation myocardial infarction. J Electrocardiol 2009; 42: 555-60. https://doi.org/10.1016/j.jelectrocard.2009.06.009 PMid:19643432 15. Karaagac K, Yontar OC, Tenekecioglu E, Vatansever F, Ozluk OA, Tutuncu A, et al. Evaluation of Tp-Te interval and Tp-Te/QTc ratio in patients with coronary artery ectasia. Int J Clin Exp Med 2014; 7: 2865-70. 16. Castro Hevia J, Antzelevitch C, Tornés Bárzaga F, Dorantes Sánchez M, Dorticós Balea F, Zayas Molina R, et al. Tpeak-Tend and Tpeak-Tend dispersion as risk factors for ventricular tachycardia/ventricular fibrillation in patients with the Brugada syndrome. J Am Coll Cardiol 2006; 47: 1828-34. https://doi.org/10.1016/j.jacc.2005.12.049 PMid:16682308 PMCid:PMC1474075 17. Henry WL, DeMaria A, Gramiak R, King DL, Kissio JA, Popp RL, et al. Report of the American Society of Echocardiography Committee on Nomenclature and Standards in two-dimensional imaging. Circulation 1980; 62: 212-7. https://doi.org/10.1161/01.CIR.62.2.212 PMid:7397962 18. Chubb H, Whitaker J, Williams SE, Head CE, Chung NA, Wright MJ, et al. Pathophysiology and management of arrhythmias associated with atrial septal defect and patent foramen ovale. Arrhythm Electrophysiol Rev 2014; 3: 168-72. https://doi.org/10.15420/aer.2014.3.3.168 PMid:26835086 PMCid:PMC4711537 19. Vecht JA, Saso S, Rao C, Dimopoulos K, Grapsa J, Terracciano CM, et al. Atrial septal defect closure is associated with a reduced prevalence of atrial tachyarrhythmia in the short to medium term: a systematic review and meta-analysis. Heart 2010; 96: 1789-97. https://doi.org/10.1136/hrt.2010.204933 PMid:20965992 20. Brouwer C, Hazekamp MG, Zeppenfeld K. Anatomical substrates and ablation of reentrant atrial and ventricular tachycardias in repaired congenital heart disease. Arrhythm Electrophysiol Rev 2016; 5: 150-60. https://doi.org/10.15420/AER.2016.19.2 PMid:27617095 PMCid:PMC5013179 21. Eryu Y, Hata T, Nagatani A, Funamoto Y, Uchida H, Fujino M, et al. Electrocardiographic RR and QT interval variability in patients with atrial septal defect and healthy children. Pediatr Cardiol 2017; 38: 582-7. https://doi.org/10.1007/s00246-016-1551-z PMid:28058479 22. Rucklova K, Koubsky K, Tomek V, Kubus P, Janousek J. Prolonged repolarization in atrial septal defect: An example of mechanoelectrical feedback due to right ventricular volume overload. Heart Rhythm 2016; 13: 1303-8. https://doi.org/10.1016/j.hrthm.2016.01.032 PMid:26829112 23. Kors JA, Ritsema van Eck HJ, van Herpen G. The meaning of the Tp-Te interval and its diagnostic value. J Electrocardiol 2008; 41: 575-80. https://doi.org/10.1016/j.jelectrocard.2008.07.030 PMid:18954608 24. Antzelevitch C, Sicouri S, Di Diego JM, Burashnikov A, Viskin S, Shimizu W. et al. Does Tpeak-Tend provide an index of transmural dispersion of repolarization? Heart Rhythm 2007; 4: 1114-6. https://doi.org/10.1016/j.hrthm.2007.05.028 PMid:17675094 PMCid:PMC1994816 25. Smetana P, Schmidt A, Zabel M, Hnatkova K, Franz M, Huber K, et al. Assessment of repolarization heterogeneity for prediction of mortality in cardiovascular disease: peak to the end of the T wave interval and nondipolar repolarization components. J Electrocardiol 2011; 44: 301-8. https://doi.org/10.1016/j.jelectrocard.2011.03.004 PMid:21511064 26. Gupta P, Patel C, Patel H, Narayanaswamy S, Malhotra B, Green JT, et al. T(p-e)/QT ratio as an index of arrhythmogenesis. J Electrocardiol 2008; 41: 567-74. https://doi.org/10.1016/j.jelectrocard.2008.07.016 PMid:18790499 27. Massin M, von Bernuth G. Clinical and haemodynamic correlates of heart rate variability in children with congenital heart disease. Eur J Pediatr 1998; 157: 967-1. https://doi.org/10.1007/s004310050979 PMid:9877033

The lake in high-altitude Kochkor region, Naryn, Kyrgyzstan. Ulan Tursunbekov, Bishkek, Kyrgyzstan.
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.Archive of Issues
AUTHOR'S CORNER
 Authors having problems with submissions please notify editor: editor@hvt-journal.com
Authors having problems with submissions please notify editor: editor@hvt-journal.com
